The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the
Question:
The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the acid-catalyzed mechanism H+ is added in the first step (see Figure). In the base-catalyzed mechanism, H+ is removed in the first step. Show the steps in the mechanism for the base-catalyzedtautomerization.
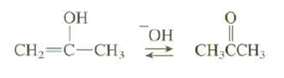
Transcribed Image Text:
ОН ОН CH=C-CH; 2 CH,CCH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (22 reviews)
CHCCH3 OH ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A strong acid or base in solution is completely a. dissociated b. neutralized c. hydrolyzed d. precipitated
-
The mechanism involved in the reaction catalyzed by phosphoglyceromutase is known to involve a phosphory-lated enzyme intermediate. If 3-phosphoglycerate is radioactively labeled with 32P, the...
-
Show the steps in the mechanism for eq. 9.18. OCH3 (9.18) CH,OH (excess) -H-H+(catalyst) OCH3
-
Given an array, Arr[] of integer numbers have size N. The task is to find out single or consecutive numbers from Arr[] with 0 sum. When found, print "1" otherwise "0". Example-1: Input: 4 (6,-2,3,-1)...
-
List some other success factors of BI.
-
The following transactions occurred in the opening and operation of Billss Delivery Service of Charlottetown: A. Bill OBrien opened the delivery service by investing $21,000 from his personal savings...
-
How does a model represent a System or Entity?
-
Presented below is an incomplete income statement and an incomplete comparative balance sheet of Cotte Corporation. Additional information:1. The receivables turnover for 2011 is 10 times.2. All...
-
Dr. Jim performed an appendectomy on Alison Thursday morning. Everything went well and Alison seemed to be recovering as expected. Saturday morning, she woke with horrible pain and an x-ray revealed...
-
After looking at the projections of the HomeNet project, you decide that they are not realistic. It is unlikely that sales will be constant over the four-year life of the project. Furthermore, other...
-
The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the product of this reaction. 1) Hg(O,CCH)2, CH3OH 2) NaBH4, NAOH CH,CH,CH CH=CH;
-
An unknown compound has the formula C6H10. (a) What is the DU for this compound? (b) When a solution of Br2 in CC14 is added to the unknown, the bromine color disappears. What information does this...
-
An early science that tried to correlate personality with measurements of parts of a persons skull is known as ________. a. Phrenology b. Psychology c. Physiology d. Personality psychology
-
What is the annual dividend on TXS preferred stock? If investors require a return of 8% on this Bock and the d dividend is payable 1 year from now, what is the price of TXS preferred stock? Suppose...
-
How firms estimate their cost of capital: The WACC for a firm is 13.00 percent. You know that the firm's cost of debt capital is 10 percent and the cost of equity capital is 20% What proportion of...
-
Many cafes are putting sensors on the bottom of cups to help alert the wait staff that a customer needs a refill. Would you introduce this technology to the cafe? If so, are there any concerns with...
-
When Sarah James husband died, she became the beneficiary of a $100,000 life insurance policy. Instead of taking the money in a lump sum, she elects to receive a monthly stipend over a period of 20...
-
A gasoline-driven and an electrically powered pump are being considered for service in a remote location. Pertinent data are as follows: The income tax rate is 35% and the acceptable return is 15%....
-
The heat engine represented by the \(P V\) diagram in Figure P21.33 exhausts \(43.5 \mathrm{~kJ}\) of thermal energy per cycle. What is the engine's efficiency? Data from Figure P21.33 P (atm) 3 2 0...
-
Determine whether the lines are parallel, perpendicular, or neither. 2x + 3y = -12, 2y - 3x = 8
-
For the reaction: K = 1.8 10 -7 at a certain temperature. If at equilibrium [O 2 ] = 0.062 M, calculate the equilibrium O 3 concentration. 30(g) 203(g)
-
Trehalose is a non-reducing disaccharide that is hydrolyzed by aqueous acid to yield 2 equivalents of D-glucose. Methylation followed by hydrolysis yields 2 equivalents of 2, 3, 4,...
-
Trehalose is cleaved by enzymes that hydrolyze a-glycosides but not by enzymes that hydrolyze -glycosides. What is the structure and systematic name of trehalose?
-
Iso trehalose and neotrehalose are chemically similar to trehalose except that neotrehalose is hydrolyzed only by -glycosidase enzymes, whereas iso trehalose is hydrolyzed by both -and-glycosidase...
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...
-
John Fleming, chief administrator for Valley View Hospital, is concerned about the costs for tests in the hospital s lab. Charges for lab tests are consistently higher at Valley View than at other...
-
A company is looking at new equipment with an installed cost of $436,859. This cost will be depreciated straight-line to zero over the project's 6-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


