The phase diagram of liquid 3 He 4 He mixtures in Figure shows that the solubility of
Question:
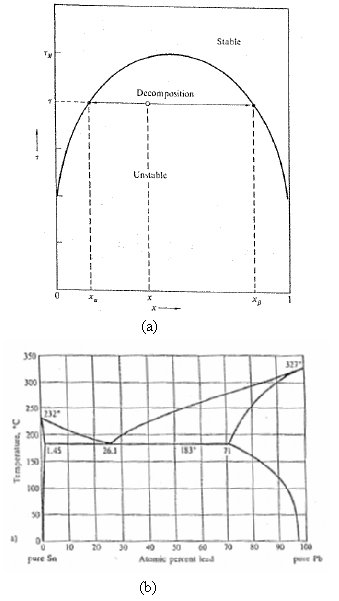
The phase diagram of liquid 3He– 4He mixtures in Figure shows that the solubility of 3He in 4He remains finite (about 6 pct) as τ → 0. Similarly, the Pb-Sn phase diagram of Figure shows a finite residual solubility of Pb in solid Sn with decreasing τ. What do such finite residual solubilities imply about the form of the function u(x)?
Transcribed Image Text:
Stabte Decomposition Unstable (a) 350 327 300 250 232 200 L.4S 190 I83 26.1 71 00 a) 20 80 10 20 30 30 90 100 pure Se Atomis peneni lead (b) Temperature, "C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The curve ux must have a shape qualitatively ...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Solid State questions
-
The phase diagram of sulfur is shown here. (a) How many triple points are there? (b) Monoclinic and rhombic are two allotropes of sulfur. Which is more stable under atmospheric conditions? (c)...
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
Use graph paper and sketch the phase diagram of oxygen, O2, from the following information: normal melting point, 218C; normal boiling point, 183C; triple point, 219C, 1.10 mmHg; critical point,...
-
Southern Stitches is a local casual clothing shop that makes a variety of t-shirt styles. Below is some actual vs. budget information for Southern's t-shirts for the month of May. Assume no...
-
This problem illustrates a deceptive way of quoting interest rates called add-on interest. Imagine that you see an advertisement for Crazy Judy's Stereo City that reads something like this: "$1,000...
-
What would you expect to happen to the price of Fastest Company's preferred shares if inflation increased and the Fed increased interest rates, with banks following suit?
-
Nicole Lutes, a partner in a regional public accounting firm, is conducting the independent technical reviewthe "cold" reviewof working papers for the December 31, 1999, year end audit of Singer...
-
Pratts Department Store is opening a new store in The Centers Mall. Customer movement tracked in its existing stores is shown below. Design a layout for Pratts new store on a 2 x 3 grid that will...
-
Describe what information you agree with, about leadership, and the parts you disagree with, about leadership From the link below...
-
Distinguish between the legal standards of gross negligence and fraud.
-
Show that the chemical potentials A and B of the two atomic species A and B of an equilibrium two phase mixture are given by the intercepts of the two-point tangent in Figure with the vertical...
-
Let B be an impurity in A, with X < < 1. In this limit the non-mixing parts of the free energy can be expressed as linear functions of x, as f0(x) = f0(0) + xf0(0), for both liquid and solid phase....
-
Gottschalk plc sponsors a defined benefit plan for its 100 employees. On January 1, 2022, the companys actuary provided the following information. Pension plan assets (fair value) ..................
-
Frances paid income tax and Class 4 NICs of 47,000 for tax year 2019-20, of which 19,000 was paid via the PAYE system. Her total liability for 2020-21 is 69,000, of which 21,000 is paid via PAYE. Her...
-
The current through an inductor of inductance \(L\) is given by \(I(t)=I_{\max } \sin (\omega t)\). (a) Derive an expression for the induced emf in the inductor as a function of time. (b) At \(t=0\),...
-
An article in the New York Post placed some of the blame on Maria Taylor for not working with Rachel Nichols after her recorded comments to come to a satisfactory resolution. What do you think?...
-
Suggest other actions the Green Bay Packer could have performed to maintain their relationships with fans and other stakeholders. Why would these other actions be suitable?
-
In some cases courts have found that landlords do have an obligation to protect tenants from criminal attack. True False
-
log 7 y 3 Write each expression as a sum of logs. Express powers as factors.
-
Write the given system without the use of matrices. D) - ()- d (x sin t + 8 (2+ 1)
-
Write the SQL code for the following: Order the reservations by class date and then by class ID. Display all fields. a. SELECT * FROM Reservations ORDER BY ClassDate, ClassID; b. DISPLAY ALL FROM...
-
One mole of an ideal gas is contained in a cylinder with a movable piston. The initial pressure, volume, and temperature are Pi, Vi, and Ti, respectively. Find the work done on the gas for the...
-
One mole of an ideal gas, initially at 300 K, is cooled at constant volume so that the final pressure is one fourth of the initial pressure. Then the gas expands at constant pressure until it reaches...
-
Continue the analysis of Problem 60 in Chapter 19. Following a collision between a large spacecraft and an asteroid, a copper disk of radius 28.0 m and thickness 1.20 m, at a temperature of 850C, is...
-
Solve the initial value problem. dy =3e3t sin (e3t-27), y(In 3)=0 dt The solution is y =
-
Solve by factoring. 5x2-16x+12=0
-
Find f-1 (x) for f (x) = 15 + 12x.

Study smarter with the SolutionInn App


