Show that the chemical potentials A and B of the two atomic species A and
Question:
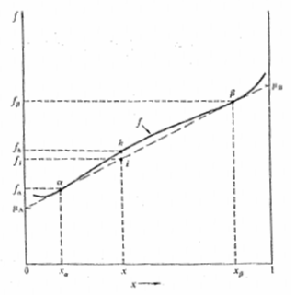
Show that the chemical potentials μA and μB of the two atomic species A and B of an equilibrium two phase mixture are given by the intercepts of the two-point tangent in Figure with the vertical edges of the diagram at x = 0 and x = 1.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
The chemical potentials for atomic species A and B are given by ...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Solid State questions
-
A space station consists of two sections A and B of equal masses, which are rigidly connected. Each section is dynamically equivalent to a homogeneous cylinder of length 15 m and radius 3 m. Knowing...
-
Chemical potential in two dimensions, show that the chemical potential of a Fermi gas in two dimensions is given by: (T) = k B T in [exp(th 2 /mk B T) 1] for n electrons per unit area. The density...
-
Given two events A and B of a sample set where P[A] = .20 P[not B] = .40 P[B|A] = .25 What is P[A or B]?
-
Wilburton Hospital is investigating the possibility of investing in new dialysis equipment. Two local manufacturers of this equipment are being considered as sources of the equipment. After-tax cash...
-
Your Christmas ski vacation was great, but it unfortunately ran a bit over budget. All is not lost: You just received an offer in the mail to transfer your $10,000 balance from your current credit...
-
Fastest Company has a debt rating of A and a tax rate of 35 percent. The current long-term government bond yield is 2 percent. Suppose the typical spread between long-term government yields and...
-
In any audit engagement, an auditor is concerned that financial statements reflect properly the economic substance of material related party transactions. To achieve this objective, auditors attempt...
-
It is time for the annual performance review of Go-Coms account executives. Account values and hours spent each week acquiring and servicing accounts are shown below. Each agent works approximately...
-
Strategic management is a popular method for running businesses which involves an analytical approach to setting goals and managing resources. Like strategic planning, strategic management often...
-
What is the comparative negligence defense? When can it shield auditors from legal liability for their actions?
-
Consider a crystal that can exist in either of two structures, denoted by and . We suppose that the x structure is the stable low temperature form and the structure is the stable high temperature...
-
The phase diagram of liquid 3 He 4 He mixtures in Figure shows that the solubility of 3 He in 4 He remains finite (about 6 pct) as 0. Similarly, the Pb-Sn phase diagram of Figure shows a finite...
-
Discuss the trade-offs involved in selecting between the two materials for each of the applications listed: (a) Sheet metal vs. reinforced plastic chairs (b) Forged vs. cast crankshafts (c) Forged...
-
The magnitude of the magnetic field in a magnetic resonance imaging (MRI) machine can be as great as \(B=3.0 \mathrm{~T}\). Under normal circumstances, this field cannot be shut off by just flipping...
-
Media and public responses create ethical demands on corporate communicators, but how and to what extent can sport organizations better support athletes and communicate about mental health?
-
South Asia has a lower income per capita than sub-Saharan Africa. Comment on the validity of this statement.
-
You are building an instrument to measure the orientation of Earth's magnetic field. Your device consists of a single conducting coil that encloses an area \(A=400 \mathrm{~mm}^{2}\) and rotates at a...
-
What instances of athlete activism have you witnessed since this chapter was published? How do they differ from Kaepernicks protest? To what degree have the maintenance of athletes reputations and/or...
-
log 5 x 4 Write each expression as a sum of logs. Express powers as factors.
-
In a certain school district, 3% of the faculty use none of their sick days in a school year. Find the probability that 5 faculty members selected at random used no sick days in a given year.
-
Write the SQL code for the following: List all the information in the patients table sorted by city. a. SELECT ALL FROM Patients ORDER BY City; b. SELECT * FROM Patients ASC City; c. SELECT * ORDER...
-
An aluminum rod 0.500 m in length and with a cross-sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 300 K. (a) If...
-
A copper ring (with mass of 25.0 g, coefficient of linear expansion of 1.70 X 10-5 (C)-1, and specific heat of 9.24 X 10-2 cal/g C) has a diameter of 5.00 cm at its temperature of 15.0C. A spherical...
-
A flow calorimeter is an apparatus used to measure the specific heat of a liquid. The technique of flow calorimetry involves measuring the temperature difference between the input and output points...
-
Convert the following ERDs to a Database schema. Follow all the steps in order as learned in this class. Identify PKs and FKs for each relation. Explain your work. Press esc to exit full screen Page...
-
How to draw a database in MS SQL Server for an entity with only a multivalued attribute and no primary key?
-
use Adventure Works Database Run each query and save the notebook with the results before submitting it. All the questions below are related to AdventureWorks database. make sure your notebook is...

Study smarter with the SolutionInn App


