The presence of a small amount of air may cause a significant reduction in the heat rate
Question:
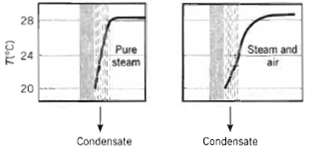
The presence of a small amount of air may cause a significant reduction in the heat rate to a water-cooled steam condenser surface. For a clean surface with pure steam and the prescribed conditions, the condensate rate is 0.020 kg/m2 ? s. With the presence of stagnant air in the steam, the condensate surface temperature drops from 28 to 24?C and the condensate rate is reduced by a factor of 2. For the air-steam mixture, determine the partial pressure of air as a function of distance from the condensate film.
Transcribed Image Text:
28 Pure steam Steam and 24 20 Condensate Condensate (3.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (21 reviews)
KNOWN Clean surface with pure steam has condensate rate of 0020 kgms for the prescribed conditions W...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Question Posted:
Students also viewed these Mechanical Engineering questions
-
As you read in the chapter, the presence of a computer network is the defining characteristic of e-business. Research the topic and define each of the following terms related to networks and...
-
A test indicates the presence of a particular disease 90% of the time when the disease is present and the presence of the disease 2% of the time when the disease is not present. If 0.5% of the...
-
In the presence of a small amount of bromine, cyclohexene undergoes the following lightpromoted reaction: (a) Propose a mechanism for this reaction. (b) Draw the structure of the rate-limiting...
-
Describe the process of testing software developed using both top-down and bottom-up development order. Which method results in the fewest resources required for testing? What types of errors are...
-
Do binary stars (double-star systems that orbit about a common center of mass) radiate gravitational waves? Why or why not?
-
The bottom chord AB in a small truss ABC (see figure) is fabricated from a W8 Ã 28 wide-flange steel section. The cross-sectional area A = 8.25 in.2 (Appendix E, Table E-1 (a)) and each of the...
-
The first law of thermodynamics basically represents the law of conservation of (a) Energy (b) Mass (c) Momentum (d) None of these.
-
1. Is the sampling method adequate? Is the attitude-measuring scale sound? Explain. 2. Using the computerized database with a statistical software package, calculate the means of the three automotive...
-
PAYROLL Using the data provided, complete the payroll calculations table. Download the deductions table from Accounting Supplemental Material Page. Submit your reconciliation in Accounting Competency...
-
Amalgamated Fenderdenter's sales are $10 million. The company spends $3.5 million for purchase of direct materials and $15 million for direct Labor, overhead is $3.5 million and profit is $500,000....
-
A spherical droplet of liquid A and radius ro evaporates into a stagnant layer of gas B. Derive an expression for the evaporation rate of species A in terms of the saturation pressure of species A,...
-
A laboratory apparatus to measure the diffusion coefficient of vapor-gas mixtures consists of a vertical, small-diameter column containing the liquid phase that evaporates into the gas flowing over...
-
Discuss the morphological differences between animal and plant development. How are they different at the cellular level? How are they similar at the genetic level?
-
What are the sociological implications of a highly bureaucratized system on the morale and motivation of public sector employees?
-
"Firstly, I would like to thank Professor Emmanuel and all my colleagues who contributed to the success of this course. Taking the Financial Modeling course improved my understanding of the financial...
-
Explain, in terms a layperson could understand, the neurobiology of drug tolerance, dependence, and withdrawal.
-
Assume Southwood Manufacturing bought three machines in a $75,000 lump-sum purchase. An independent appraiser valued the machines as follows: Machine Number Appraised Value 1 $26,000 2 43,000 3...
-
Restoration Company has the following inventory transactions for January: Opening Inventory January 1st $32,000 (4,000 units @ $8) Purchase #1 January 10th 9,200 units @ $11 Purchase #2...
-
A portfolio consists of 75% invested in Security A with an expected return of 35% and 25% invested in Security B with an expected return of 7%. Compute the expected return on the portfolio.
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
A sample of nitrogen gas in a 1.75-L container exerts a pressure of 1.35 atm at 25 C. What is the pressure if the volume of the container is maintained constant and the temperature is raised to 355 C?
-
A static service music wire helical compression spring is needed to support a 20-lbf load after being compressed 2 in. The solid height of the spring cannot exceed 1 in. The free length must not...
-
Not all springs are made in a conventional way. Consider the special steel spring in the illustration. (a) Find the pitch, solid height, and number of active turns. (b) Find the spring rate. Assume...
-
A holding fixture for a work piece 1 in thick at clamp locations is being designed. The detail of one of the clamps is shown in the figure. A spring is required to drive the clamp upward while...
-
Dollar-Value LIFO On January 1, 2018, Sato Company adopted the dollar-value LIFO method of inventory costing. Sato's ending inventory records appear as follows: Year Current Cost Index 2018 $31,600...
-
How many standards are included in the ELP accreditation standards? What stands out to you about the different areas? What are the topic areas under administration and leadership? What are the...
-
What is performance management systems? Do the companies where you (or others you know) have worked used performance management systems rather than performance appraisal systems? If yes, what kind of...

Study smarter with the SolutionInn App


