The Thomson model of a hydrogen atom is a sphere of positive charge with an electron (a
Question:
The Thomson model of a hydrogen atom is a sphere of positive charge with an electron (a point charge) at its center. The total positive charge equals the electronic charge e. Prove that when the electron is at a distance r from the center of the sphere of positive charge, it is attracted with a force where R is the radius of thesphere.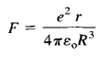
,3 4те,R
Step by Step Answer:
R F eE ...View the full answer



Related Video
In Physics, a force is an influence that can change the motion of an object. A force can cause an object with mass to change its velocity, i.e., to accelerate. Force can also be described intuitively as a push or a pull. A force has both magnitude and direction, making it a vector quantity. In this video, the concept of force and torque is demonstrated with the practical application of changing the flat tire of the car.
Students also viewed these Electrodynamics questions
-
The nucleus of a hydrogen atom is a single proton, which has a radius of about 1.0 x 10-15 m. The single electron in a hydrogen atom normally orbits the nucleus at a distance of 5.3 x 10-11 m. What...
-
The electron in a hydrogen atom is in the first excited state, when the electron acquires an additional 2.86 eV of energy. What is the quantum number n of the state into which the electron moves?
-
The electron of a hydrogen atom is in the n = 2 state. The electron makes a transition to the ground state. (a) What is the energy of the photon according to the Bohr model? (b) The linear momentum...
-
Being able to identify an industry to use for benchmarking yourfirm's results with similar companies is frequently not easy. Choose a type of business and go to www.naics.com. This Web site allows...
-
Consider Figure 15-7. Describe the basic shape that this figure would take if the Fed had instead generated a multiple contraction in the money supply by removing $100,000 in reserves from the...
-
Flintstone Children's Vitamins produces multivitamins for children. Currently a bottle of 100 vitamins sells for $8.00. This multivitamin contains many beneficial vitamins such as Vitamin A and...
-
Define outsourcing and offshoring. Compare and contrast the two as HR administrative tools. Give examples of the decision factors to consider when choosing one over the other.
-
Income statement data for Grendel Images Company for the years ended December 31, 2010 and 2009 are as follows: (a) Prepare a comparative income statement with horizontal analysis, indicating the...
-
List the characteristics (seven) of insurance contracts and explain each characteristic of insurance contract. 4. List and briefly explain the types of insurers as classified by place of...
-
On January 1, 2014, Prince Corporation acquired 70% of the 100,000 outstanding voting shares of Song Limited for a cash consideration of $1,015,000. On that date, shares of Song Limited were trading...
-
In a certain region, the electric field is given by D = 2(z + l)cos a (z + l)sin a + 2 cos az C/m2 (a) Find the charge density. (b) Calculate the total charge enclosed by the volume 0 < < 2, 0 < <...
-
Three concentric spherical shells r = 1, r = 2, and r = 3 m, respectively, have charge distributions 2, - 4, and 5 C/m2. (a) Calculate the flux through r = 1.5 m and r = 2.5 m. (b) Find D at r = 0.5,...
-
Why do firms issue convertible bonds and bonds with warrants?
-
Identify three items of nontaxable receipts which nevertheless increase earnings and profits.
-
Identify and briefly describe the seven types of corporate reorganization.
-
What is meant by a "status of liquidation"?
-
What limits are imposed on the medical expense deduction?
-
What is E&P and what is its significance?
-
Set out Loewensteins visceral factor model. What are the advantages of Loewensteinss approach versus Damasios somatic marker hypothesis versus other analyses of emotion in economic decision-making?
-
In exchange for land, the company received a 12-month note on January 1. The face amount of the note is $1,000, and the stated rate of interest is 13%, compounded annually. The 13% rate is equal to...
-
From the outcome of the transformation shown in Fig. P6.48, indicate whether the levorotatory enantiomer of the product has the R or S configuration. Draw a structure of the product that shows its...
-
Describe the components of the capital account.
-
Which balance-of-payments account would be associated with each of the following items? a. An export of goods b. A purchase of bonds c. A gift to someone in another country d. A dividend paid from...
-
Balance-of-payments accounting always involves double entries because the inflow of goods and services is always accompanied by financial outflows and vice versa. For each of the following scenarios,...
-
A retirement home in Florida costs $ 2 0 0 , 0 0 0 today. Housing prices in Florida are increasing at a rate of 4 % per year. Joe wants to buy the home in 8 years when he retires. Joe has $ 2 5 , 0 0...
-
1. (55 points) Answer the following questions about the steady state system shown below. (Don't worry about starting from 1st Law - use common engineering assumptions.) a) Is it appropriate to use...
-
Describe at least three ways that Guidance Software responds to and helps to manage changes in our technology - driven world? Describe what forecasting involves and list three limitations to...

Study smarter with the SolutionInn App


