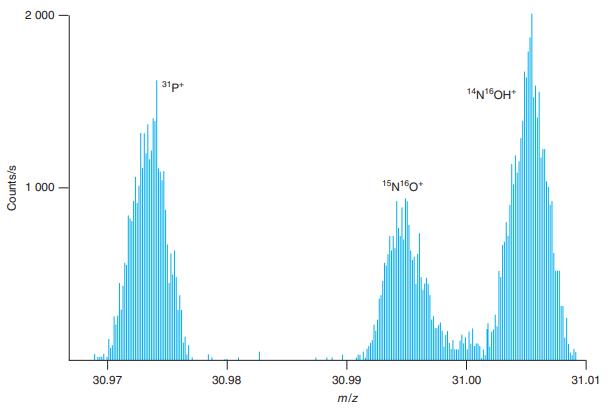
The two peaks near m/z 31.00 in Figure 21-9 differ in mass by 0.010 Da. Estimate the
Question:
The two peaks near m/z 31.00 in Figure 21-9 differ in mass by 0.010 Da. Estimate the resolving power of the spectrometer from the expression m/∆m without making any measurements in the figure.
Figure 21-9
Transcribed Image Text:
2 000 31p 14N1OH* 1 000 15N10* 30.97 30.98 30.99 31.00 31.01 m/z Counts/s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The overlap at the base of ...View the full answer

Answered By

Sufiyan Ahmed Tariq
I am a Chartered Accountant and an Associate Public & Finance Accountant. I also hold a bachelors of Commerce degree. I have over 8 years of experience in accounting, finance and auditing. Through out my career, I have worked with many leading multinational organisation.
I have helped a number of students in studies by teaching them key concepts of subjects like accounting, finance, corporate law and auditing. I help students understanding the complex situation by providing them daily life examples.
I can help you in the following subject / areas:
a) Accounting;
b) Finance;
c) Commerce;
d) Auditing; and
e) Corporate Law.
4.90+
7+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Which of these compounds has two strong peaks near 1520 and 1350 cm1 in its IR spectrum?
-
FIGURE CP33.73 shows two nearly overlapped intensity peaks of the sort you might produce with a diffraction grating (see Figure 33.9b). As a practical matter, two peaks can just barely be resolved if...
-
In Figure horizontal scaffold 2, with uniform mass m2 = 30.0 kg and length L2 = 2.00 m, hangs from horizontal scaffold 1, with uniform mass m1 = 50.0 kg. A 20.0 kg box of nails lies on scaffold Z,...
-
Consider three (ideally other) countries for which evidence features here. What are the key influences on cross-national comparative variation in the place and role of the HRM function?
-
Weber Limited is trying to determine the value of its ending inventory at February 28, 2014, the companys year-end. The accountant counted everything that was in the warehouse as of February 28,...
-
The current gain for the transistor in the circuit in Figure P5.47 is \(\beta=60\). Determine \(R_{B}\) such that \(V_{O}=8.8 \mathrm{~V}\) when \(V_{I}=5 \mathrm{~V}\) and \(I_{C} / I_{B}=25\). RB...
-
Requirements 1. Using the following selected accounts of Juba Electrical, Inc., at September 30, 2010, prepare the entitys closing entries: 2. What is Juba Electricals ending Retained Earnings...
-
Refer to the financial statements of Tootsie Roll Industries and the Notes to Consolidated Financial Statements in Appendix A. Instructions Answer the following questions. (a) What were Tootsie Rolls...
-
What is important to be succesfullin usingEthical Decision Criteria and what might bedeveloped in encouraging people in decision making?
-
When set at the standard position, Autopitch can throw hard balls toward a batter at an average speed of 60 mph. Autopitch devices are made for both major- and minor-league teams to help them improve...
-
Measure the width at half-height of the tallest peak in the spectrum below and calculate the resolving power of the spectrometer from the expression m/m 1/2 . Would you expect to be able to...
-
The highest resolution mass spectra are obtained by Fourier transform ion cyclotron resonance mass spectrometry. Molecular ions of two peptides (chains of seven amino acids) differing in mass by...
-
You have been asked to evaluate the profitability of building a new distribution center under the following conditions: The proposal is for a distribution center costing $1,500,000. The facility has...
-
Explain the terms : state, phase, process and cyclic process.
-
Define the term heat.
-
What is PMM-1.
-
State zeroth law of thermodynamics.
-
Explain the concept of temperature.
-
In Exercises use the integration capabilities of a graphing utility to approximate the work done by a press in a manufacturing process. A model for the variable force F (in pounds) and the distance x...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
A mixture containing only aluminum tetrafl uoroborate, Al(BF 4 ) 3 (FM 287.39), and magnesium nitrate, Mg(NO 3 ) 2 (FM 148.31), weighed 0.282 8 g. It was dissolved in 1 wt% HF(aq) and treated with...
-
Explain what is done in thermogravimetric analysis.
-
Why is it less desirable to wash AgCl precipitate with aqueous NaNO 3 than with HNO 3 solution?
-
based on the article How Chili's Is Prepping for Tough Times, Starting With the Fries by Heather Haddon. What is corporate social responsibility, and what is one way that Chili's can better pursue...
-
A rocket takes off vertically from the Launchpad with no initial velocity but a constant upward acceleration of 2.25 m/s. At 15.4 s after blastoff, the engines fail completely so the only force on...
-
At the beginning of year 1, a company acquires a $1,000,000 term loan with an interest rate of 7.50% and an annual payment based on a 15-year amortization. The total liabilities section of the...

Study smarter with the SolutionInn App


