Thiamin, or vitamin Bi, contains a positively charged five-membered nitrogen?sulfur heterocycle called a thiazolium ring. Explain why
Question:
Thiamin, or vitamin Bi, contains a positively charged five-membered nitrogen?sulfur heterocycle called a thiazolium ring. Explain why the thiazolium ring is aromatic
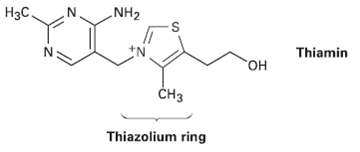
Transcribed Image Text:
NH2 Нас. Thiamin но, сHз Thiazolium ring
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
H3C H OH The heterocyclic thiazolium ring contains sixx ...View the full answer

Answered By

Milan Mondal
I am milan mondal have done my Msc in physics (special astrophysics and relativity) from the University of burdwan and Bed in physical science from the same University.
From 2018 I am working as pgt physics teacher in kendriya vidyalaya no2 kharagpur ,west bengal. And also I am doing advanced physics expert in chegg.com .also I teach Bsc physics .
I love to teach physics and acience.
If you give me a chance I will give my best to you.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why the systems approach is necessary to manage projects.
-
Explain why the following statement is true: Other things being equal, firms with relatively stable sales are able to carry relatively high debt/assets ratios.
-
Explain why the insurance industry is regulated.
-
Prepare journal entries to record each of the following sales transactions for the sales company. The company uses a perpetual inventory system and the gross method. 1. April 1st Sold for $3,000,...
-
What should employers consider when looking to hire veterans?
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
How does an auditor test whether recorded accounts receivable exist?
-
Adria Lopez is considering the purchase of equipment for Success Systems that would allow the company to add a new product to its computer furniture line. The equipment is expected to cost $ 300,000...
-
Kitty is paid $1200 weekly, also receives the following in addition to her regular salary each pay: a group term life insurance non-cash taxable benefit of $15 and a taxable cash car allowance of...
-
What are the major causes of information risk? How can information risk be reduced?
-
Draw an orbital picture of furan to show how the molecule isaromatic. Furan :O:
-
Show the relative energy levels of the seven molecular orbitals of the Cycloheptatrienyl system. Tell which of the seven orbitals are filled in the cation, radical, and anion, and account for the...
-
The double-toggle mechanism shown is used in a punching machine. Knowing that links AB and BC are each of length 6 in., determine the couple M required to hold the system in equilibrium when Ï =...
-
1). An audio speaker producing a steady sound at an outdoor concert is 28 ft away from you. If you move to a position where the speaker is 64 ft distant, by what factor will the amplitude of the...
-
Restaurant furnishings with an original cost of $38,000 and accumulated depreciation account of $32,000, was sold for $4,200 cash. How will the proceeds of the sale be reported in a SCF, indirect...
-
DDD Ltd. is a private corporation with a taxation year ending on December 31. The sole shareholder is Donald Darwin Dorsey who is a Canadian resident throughout 2023. In 2023, DDD Ltd. had the...
-
1) Livingstone's Warehouse received an invoice dated July 17 for $1,200, with terms of 2/10, 1/15, n/60. On July 20, Livingston's Warehouse sent a partial payment of $650. How much will be credited...
-
1). A person stands directly in front of two speakers that are emitting the same pure tone. The person then moves to one side until no sound is heard. At that point, the person is 7.15 m from one of...
-
If the pion slows down to \(0.99990 c\), about what percentage of its kinetic energy is lost? A. \(0.03 \%\) B. \(0.3 \%\) C. \(3 \%\) D. \(30 \%\)
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Why are the concentrations of solids and liquids omitted from equilibrium expressions?
-
On treatment with phenylhydrazine, aldohexoses A and B give the same osazone. On treatment with warm nitric acid, A gives an optically inactive aldaric acid, but sugar B gives an optically active...
-
In 1891, Emil Fischer determined the structures of glucose and the seven other D-aldohexoses using only simple chemical reactions and clever reasoning about stereochemistry and symmetry. He received...
-
Draw and name the enantiomers of the sugars shown in Figure 23-2. Give the relative configuration (D or L) and the sign of the rotation in each case.
-
es Hart, Attorney at Law, experienced the following transactions in Year 1, the first year of operations: 1. Accepted $16,600 on April 1, Year 1, as a retainer for services to be performed evenly...
-
Dahlia Corporation has a current accounts receivable balance of $439,516. Credit sales for the year just ended were $5,503,810. a. What is the receivables turnover? Note: Do not round intermediate...
-
Why does the organizational structure hold political significance? Provide an in-depth analysis of this concept using examples from both academic literature and real-world instances. Additionally,...

Study smarter with the SolutionInn App


