This hydroboration reaction forms two products. Show these products and explain which one you expect to be
Question:
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor.
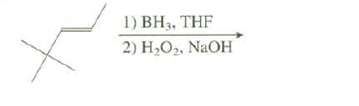
Transcribed Image Text:
1) BH3, THF 2) H,O2. NAOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The right alcohol is the ...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Which alkyl halide would you expect to be more reactive in an SN2 reaction with a given nucleophile? In each case, you can assume that both alkyl halides have the same stability a. CH3CH2CH2Br or...
-
Which alkyl halide would you expect to be more reactive in an SNI solvolysis reaction? CH2CH3 CH3 or - CH CHCH H CH CH2CH H
-
Compiler Error says main must return int. Why? I just need tomake this code work. Thats all. COMPILER :: gmichel@ubuntu:~/Desktop/CS302REDO/Project6$ g++ -Wall-std=c++11 pro6.cpp -o project...
-
Management accountants need a wide variety of skills for their roles in organizations. These skills can be classified as either technical or nontechnical. Requirement For each of the following...
-
Explain how tolerance limits to environmental factors determine distribution of a highly specialized species such as the saguaro cactus.
-
Who is responsible for performing DT&E?
-
Mancuso Corporation amended its pension plan on January 1, 2010, and granted $160,000 of prior service costs to its employees. The employees are expected to provide 2,000 service years in the future,...
-
The following selected transactions relate to liabilities of Pacific Coast Adventures. Pacific Coast's fiscal year ends on December 31. January 13 Negotiate a revolving credit agreement with First...
-
Consider the language S*, where S = {aa b}. How many words does this language have of length 4? of length 5? of length 6? What can be said in general?
-
Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
-
Show preparation of these alcohols fromalkenes. b) c)
-
Refer to the year-end financial statements and accompanying notes of Shoppers Drug Mart (SDM). Instructions Using ratio and other analyses, prepare an assessment of SDM's solvency and financial...
-
The Hernys law contant for nitrogen dioxide is k H = 4.0x10 -2 M/atm at 25C. What is the solubility of NO 2 at 25C in water at a partial pressure of 575 mmHg?
-
A truck driver averages 92 kilometers per hour between 2 p.m. and 6 p.m. Can you determine the speed of the truck at 4 p.m.?
-
You are buying a car, and have settled on a price of $22,678.95. You put $2,678.95 down and borrow the rest at an 8.5% APR, compounded monthly for 48 months. This requires a monthly payment of...
-
The total column mass of water (vapor + liquid) in the atmosphere is observed to be, on average, about 25 kg m 2 (99.5% of which is in the gas phase) (a) What equivalent depth of liquid water does...
-
Lets say that 88% of all business start-ups in the IT industry report that they generate a profit in their first year. If a sample of 15 new IT business start-ups is selected, find the probability...
-
A source adds \(150 \mathrm{~J}\) of thermal energy to a steady device while \(275 \mathrm{~J}\) of work is done on the device. The device is in contact with its environment. Without specifying...
-
Show, if u(x, y) and v(x, y) are harmonic functions, that u + v must be a harmonic function but that uv need not be a harmonic function. Is e"e" a harmonic function?
-
Calculate the pH after 0.10 mole of NaOH is added to 1.00 L of the solution in Exercise 32, and calculate the pH after 0.20 mole of HCl is added to 1.00 L of the solution in Exercise 32. Data in...
-
Fill in the missing reagents a?d in the following synthesis of racemic methamphetamine from benzene. b, c NHCH3 (R,S)-Methamphetamine
-
How might a reductive amination be used to synthesize ephedrine, an amino alcohol that is widely used for the treatment of bronchialasthma? CHCHNHCH3 Ephedrine H3
-
One problem with reductive amination as a method of amine synthesis is that by-products arc sometimes obtained. For example, reductive amination of benzaldehyde with methylamine leads to a mixture of...
-
How does the perception of organizational justice affect employee motivation and job satisfaction, and what can organizations do to promote a sense of fairness and equity ?
-
What factors contribute to the formation of high-performance teams, and how can organizations cultivate and sustain these dynamics over time ?
-
The standard 2-pass spur gear train with the # of teeth on each gear as shown is driven is driven at 1000 RPM from shaft 2 with a torque of 25 in-lb. All the gears in the set have a diametral pitch...

Study smarter with the SolutionInn App


