Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,,
Question:
Show the products of thesereactions:
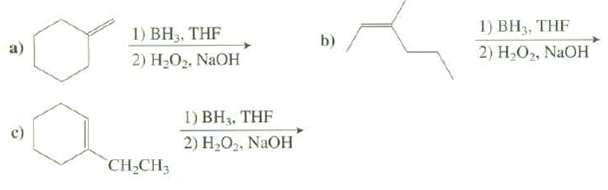
Transcribed Image Text:
1) BH3, THF 1) BH3. THF 2) Н.О. NaOH b) 2) Н,О, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a OH o b ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
As CEO of Aqua Marine, Carrie Easton knows it is important to control costs and to respond quickly to changes in the highly competitive boat-building industry. When Rose Consulting proposes that Aqua...
-
Which wavelengths do our eyes respond to, and why? (Refer to fig. 2.13.) About how long are short ultraviolet wavelengths compared to microwave lengths?
-
What is OT&E?
-
Dillons Corporation made credit sales of $30,000 which are subject to 6% sales tax. The corporation also made cash sales which totaled $20,670 including the 6% sales tax. (a) Prepare the entry to...
-
How does an organization's approach to work-life balance influence employees' loyalty and long-term commitment ?
-
During bad economic times, industrial workers are dismissed and are often replaced by machines. The history of 100 workers whose loss of employment is attributable to technological advances is...
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Information about Lindas Boards is presented in E6-4. Additional data regarding Lindas sales of Xpert snowboards are provided below. Assume that Lindas uses a perpetual inventory system. In E6-4,...
-
Describe the Indian Major Crimes Act. Explain how there came to be a boundary between tribal and federal law. Discuss the ruling in McGirt v. Oklahoma . Do you agree with the courts decision? Explain...
-
Given that f(x)=-6 and g(x)=-7x+5, find if it exists.
-
How do emergent forms of organizational structures, like holacracy or networked organizations, challenge traditional paradigms and foster dynamic collaboration and creativity?
-
45% of a baker's total expenses are for the cost of fuel and the remainder is for the cost of material. a) in 1989, the baker's total expenses were $7000, calculate the cost of his material in 1989,...
-
A project costs Birr 2,000,000 and yields annually a profit of Birr 300,000 after depreciation at 12.5% but before tax at 50%. Calculate the payback period.
-
For the steady device shown in Figure P21.4, determine the values of \(W_{\text {in }}\) and \(Q_{\text {in }}\). Data from Figure P21.4 W = ? Qin = ? W out=320J
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
Calculate the mass of sodium acetate that must be added to 500.0 mL of 0.200 M acetic acid to form a pH = 5.00 buffer solution.
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH 3,5-Dimethylisoxazole
-
Account for the fact that p-nitro aniline (pKa = 1.0) is less basic than m-nitro aniline (pKa = 2.5) by a factor of 30. Draw resonance structures to support your argument. (The PKa values refer to...
-
1.Taxpayer, a cash method, calendar year taxpayer, engaged in the following transactions in shares of stock. Consider the amount and character of T's gain or loss in each transaction: (a)T bought 100...
-
where company shows it uneanedd revuene and how it is decribedd in the footnotes?
-
Required Compute variances for the following items and indicate whether each variance is favorable (F) or unfavorable (U): Note: Select "None" if there is no effect (i.e., zero variance). Item Sales...

Study smarter with the SolutionInn App


