Trans-Decalin is more stable than its cis isomer, but cis-bicyclo [4.1.0] heptane is more stable than its
Question:
Trans-Decalin is more stable than its cis isomer, but cis-bicyclo [4.1.0] heptane is more stable than its transisomer.Explain.
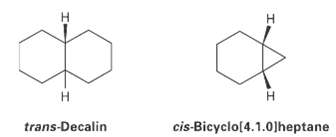
Transcribed Image Text:
trans-Decalin cis-Bicyclo[4.1.0]heptane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
Build models to see the stability difference between the two 410 rin...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Normally, a Trans alkene is more stable than its cis isomer Trans-Cyclooctene, however, is less stable than cis-Cyclooctene by 38.5kJ/mol. Explain.
-
Trans-2-Buterw is more stable than cic-2-hutene by only 4kJ/mol, but trans-2, 2, 5; 5-tetramethyl-3-hexene is more stable than its cis isomer by 3kJ/mol. Explain.
-
1-Methylcyclohexene is more stable than methylene-cyclohexane (A, in the margin), but methylene-cyclopropane (B) is more stable than 1-methyl-cyclopropene. Explain.
-
If a natural disaster, such as the 2010 drought in Russia, hits food production, use supply and demand analysis to figure out how this affects consumers and producers. Does everyone lose or are some...
-
1. What are the ways that Shelby and Mark might earn money from stock investments? What are the risks involved? 2. If Shelly and Mark want to use the Internet to evaluate stocks, what are four Web...
-
A piston cylinder device of volume 1 m3 contains 3 kg of water. The piston, which has an area of 100 cm2, exerts a force of 1.7 kN on the pin to keep it stationary. Determine (a) The temperature. (b)...
-
Define precision and reliability statistically and indicate their meaning in auditing.
-
EOQ, uncertainty, safety stock, reorder point. Chadwick Shoe Co. produces and sells an excellent quality walking shoe. After production, the shoes are distributed to 20 warehouses around the country....
-
How loud is too loud? Loudness can definitely be subjective to the listener, but there are also quantifiable numbers to reference loudness. There are decibel levels that have been proven to cause...
-
This is really an odd situation, said Jim Carter, general manager of Highland Publishing Company. We get most of the jobs we bid on that require a lot of press time in the Printing Department, yet...
-
Using molecular models as well as structural drawings explain why trans-ans decalin is rigid and cannot ring-flip, whereas cis-decalin cans easily ring-flip.
-
Myo-lnositol, one of the isomers of 1, 2, 3, 4, 5, 6-hexahydroxvcyclohexane, acts as a growth factor in both animals and microorganisms. Draw the most stable chair conformation ofmyo-inositol. . " ....
-
Which of the following molecules may be polar? (i) pyridine, (ii) nitroethane, (iii) gas-phase HgBr 2 , (iv) B 3 N 3 H 6 .
-
3. On December 31, 2019, the van was sold for $3,000 cash. Compute the amount of the gain or loss on the sale and prepare the journal entry to record the sale using: (a) straight-line method 2020 3....
-
In 2 0 2 2 , Emma wrote a play called " My Kitchen Antics" which was based on the life of her late mother who was a celebrity chef. In August 2 0 2 2 she was offered a contract by a Sydney Theatre...
-
Calculate the net income for Green Thumb Company. Then, assuming the company had retained earnings of $ 1 6 2 , 0 0 0 as of January 1 , 2 0 2 0 , and paid out $ 4 6 , 0 0 0 in dividends during the...
-
Years ago, February Frost Inc. ( FFI ) borrowed $ 6 7 5 , 0 0 0 from Sweetheart Corporation ( Sweet ) in the form of a 1 0 year, 1 0 % note due on December 3 1 , 2 0 2 4 . FFI has had declining sales...
-
Consider a two-period endowment economy. There are two countries, France and Morocco. Consumers in France have log preferences: In C + In C2 where C1 and C2 refer to French consumption in period 1...
-
Consider the extraction process of Problem 7.3. Calculate the total amount of solvent required if the extraction is done in a crosscurrent cascade consisting of 5 ideal stages. Use equation (3-89)...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Compare the critical temperatures of NH 3 and N 2 (Table 12.6). Which gas has the stronger intermolecular forces? Table 12.6 TABLE 12.6 Some Critical Temperatures, T, and Critical Pressures, P...
-
From each set of resonance structures that follow, designate the one that would contribute most to the hybrid and explain your choice: (a) (b) (c) (d) (e) (f) HO HO
-
Give the structures of the products that would be formed when 1,3-butadiene reacts with each of the following: (a) (b) (c) (d) OMe OMe CN CN
-
Cyclopentadiene undergoes a Diels-Alder reaction with ethene at 160-180(C. Write the structure of the product of this reaction.
-
Go to: https://www.instagram.com/ryderseyewear/ on your desktop, laptop, or mobile (or a combination of all 3). You are the new Social Media Marketing Manager for Ryders Eyewear. You've been asked...
-
The current price of one share of stock is 70.00. The stock pays no dividends. The risk-free rate is 3%. One year call and put options on the stock are available for various strike prices. Mr. Smith...
-
> Characterize the marketplace. o Size, growth, demographics, structure, competition o Research the market and give an idea about each point > Describe the content of the business website > Conduct a...

Study smarter with the SolutionInn App


