Treatment of an ?, ? unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The
Question:
Treatment of an ?, ? unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The reaction is specific to unsaturated ketones; isolated alkene double bonds do not react. Propose a mechanism.
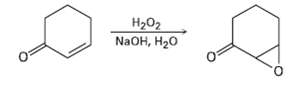
На02 NaOH, H20 O.
Step by Step Answer:
HOOH OH Hydrogen peroxide and hydroxide react to form water and ...View the full answer



Related Video
Hydrogen peroxide can be used as a mild antiseptic to curb superficial skin infections such as athlete’s foot, but only in diluted quantities. To combat stinky feet, try soaking your feet in a solution of 1 part 3% hydrogen peroxide and 3 parts warm water for 15-20 minutes, then drying them thoroughly. This will kill odor-causing bacteria and soften your feet. To treat athlete\'s foot, you can use a similar solution, but only in diluted quantities, and soak your feet for 30 minutes. Hydrogen peroxide can also be used to keep vegetables fresh by adding 1/4 cup to a bowl of cold water, soaking the vegetables for 20-30 minutes, then draining, drying, and refrigerating them. Alternatively, you can spray vegetables with a solution of 3% hydrogen peroxide and let them stand for a few minutes before rinsing and drying. To keep leftover salad fresh, spray it with a solution of 1/2 cup water and 1 Tbsp. 3% hydrogen peroxide, drain, cover, and refrigerate.
Students also viewed these Organic Chemistry questions
-
Hydrogen peroxide in aqueous solution decomposes by a first-order reaction to water and oxygen. The rate constant for this decomposition is 7.40 104/s. What quantity of heat energy is initially...
-
Hydrogen peroxide and ferrous sulfate react to produce hydroxyl radical (HO), as reported in 1894 by English chemist H. J. H. Fenton. When tert-butyl alcohol is treated with HO generated this way, it...
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Write a detailed executive summary about the appropriation and advancement of Information and Communication Technology (ICT)
-
Groupthink is the tendency for teams to put such a high premium on agreement that directly (or indirectly) punish dissent. Was the Schumacker's team guilty of groupthink? Why or why not?
-
Four people invested in a restaurant. One person invested $100,000. Two others invested in the ratio x:2x, and the fourth person invested an amount equal to the other three investors combined. The...
-
When is DT&E performed during the System Development Phase?
-
Prepare the journal entries to record the following transactions on Derrick Companys books using a perpetual inventory system. (a) On March 2, Derrick Company sold $900,000 of merchandise to Rose...
-
Answer the questions in the picture below using all listed information 15. Ugh Kinetic Elastic 200 ZE 2 kg 1 m 2 kg 0.5 A 2 kg mass is pushed 0.5 m into a spring with spring constant 200 N/m on a...
-
Rialto Group received a 100,000 grant from the government to acquire 500,000 of delivery equipment on January 2, 2022. The delivery equipment has a useful life of 5 years. Rialto uses the...
-
Tran?s alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine. Propose a mechanism for the epoxide ? alkene reaction. ...
-
One of the biological pathways by which an amine is converted to a ketone involves two steps: (1) oxidation of the amine by NAD+ to give an imine, and (2) hydrolysis of the imine to give a ketone...
-
The CEO of Sweet Snackfoods is concerned with the amount of resources currently spent on customer warranty claims. Each box of snacks is printed with the guarantee: Satisfaction guaranteed or your...
-
Williams Group started a new business on September 1, 2021. That same day, Williams Group also received $430,000 of cash from issuing common stock and borrowed $230,000 from a friend. On September 1,...
-
Matt loves cupcakes (C) and hates bananas (B). However, Matt is always willing to eat one more banana as long as he gets to eat two more cupcakes. Carefully draw two indifference curves for Matt and...
-
Analyse effective leadership strategies for enhancing performance in public enterprises. Use practical examples from the Namibian corporate sector to illustrate your answers.
-
Why do some manufacturing companies offer extended warranties to their customers if they must record a liability on their Balance Sheet for these warranties?
-
Riverwood Accounting Company has the following account in its cost records: Work in Process Jones Audit Direct labor 2 0 0 , 0 0 0 Services completed 4 2 2 , 5 3 0 Project overhead 2 7 0 , 0 0 0...
-
Our analysis of the hydrogen atom simply extends to any element which has been ionized to have a single electron orbiting the nucleus. In this problem, we will consider such an atom, whose nucleus...
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Use data from Table 10.3 to estimate the enthalpy change ( r H) for the following reaction. Table 10.3 CH6(g) + Cl2(g) CH5Cl(g) + HCl(g) AH = ?
-
Show how you might prepare aniline from each of the following compounds: (a) Benzene (b) Bromobenzene (c) Benzamide
-
Show how you might synthesize each of the following compounds from 1-butanol: (a) Butylamine (free of 28 and 38 amines) (b) Pentylamine (c) Propylamine (d) Butylmethylamine
-
Show how you might convert aniline into each of the following compounds. (You need not repeat steps carried out in earlier parts of this problem.) (a) Acetanilide (b) N-Phenylphthalimide (c)...
-
Inflation in the United States is 2% per year and in Australia, it is 3% per year. The nominal interest rate in Australia is 6%. What does the Fisher effect predict the nominal interest rate in the...
-
Johnny's Lunches is considering purchasing a new, energy-efficient grill. The grill will cost $31,000 and will be depreciated straight-line over 10 years to a salvage value of zero. The grill will...
-
John wants to "roll in" or finance the loan fee of $3,800 into the loan amount which would make the loan $90,800 and the interest rate is 6%. Assume that the lender agrees to allow the loan fees to...

Study smarter with the SolutionInn App


