Upon consideration of the SiO2???Al2O3 phase diagram, Figure, for each pair of the following list of compositions,
Question:
Upon consideration of the SiO2???Al2O3 phase diagram, Figure, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices.
(a) 20 wt% Al2O3-80 wt% SiO2 and 25 wt% Al2O3-75 wt% SiO2
(b) 70 wt% Al2O3-30 wt% SiO2 and 80 wt% Al2O3-20 wt%SiO2
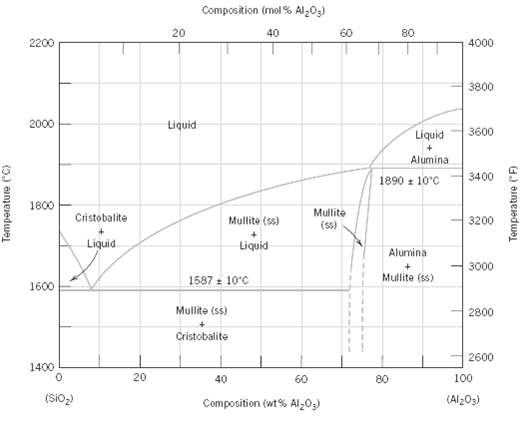
Transcribed Image Text:
Composition (mol % Al203) 80 20 40 60 2200 4000 3800 2000 Liquid Liquid 3600 Alumina 3400 1890 + 10°C 1800 Mullite (s) Cristobalite 3200 Mullite (ss) Liquid Liquid Alumina 3000 Mullite (ss) 1587 : 10°C 1600 Mulite (ss) 2800 Cristobalite 2600 1400 20 100 40 60 80 (SIO,) (Al203) Composition (wt % Al;03) Temperature ("C) Temperature ("F)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
a The 25 wt Al 2 O 3 75 wt SiO 2 will be more desirable because ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
For each pair of the following molecules, indicate whether its members are identical, structural isomers, conformers, or stereoisomers. How would you describe the relation between conformations when...
-
Upon consideration of the SiO2-Al2O3 phase diagram, Figure 12.27, for each pair of the following list of compositions, which would you judge to be the more desirable refractory? Justify your choices....
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
Marsden Corp has developed a new strategic plan after rushing defective products to market hurt the company's sales and image. The strategic plan's initiatives are to focus on quality and develop...
-
Briefly describe the Religion of the Iceland. Briefly describe the Government of the Iceland. Briefly describe the Technology of the Iceland. How do these cultural elements described in this peer...
-
Identified various issues and/or obstacles related to convergence beween GAAP and iGAAP. Choose one such issue and address the following: Briefly summarize the issue. What makes this issue...
-
As velocity increases, the propeller blades relative wind moves in relation to the chord line. Does the AOA increase or decrease? What effect does this have on propeller RPM?
-
Refer to P&Gs financial statements and accompanying notes to answer the following questions. (a) Under P&Gs stock-based compensation plan, stock options are granted annually to key managers and...
-
Distribution is a statistical concept used in data research. Those seeking to identify the outcomes and probabilities of a particular study will chart measurable data points from a data set,...
-
Michael Marrioni is in the payroll department at Seven Wonders International Foods. He is part of a social organization, along with several of his coworkers. Which of the following ethical standards...
-
Find the maximum temperature to which the following two magnesia???alumina refractory materials may be heated before a liquid phase will appear. (a) A spinel-bonded alumina material of composition 95...
-
Compute the mass fractions of liquid in the following refractory materials at 1600C (2910F): (a) 6 wt% Al2O3-94 wt% SiO2 (b) 10 wt% Al2O3-90 wt% SiO2 (c) 30 wt% Al2O3-70 wt% SiO2 (d) 80 wt% Al2O3-20...
-
What was the point of the organizational changes made by Cor Boonstra? What was he trying to achieve? Do you agree with Frans van Houten's decision to keep the same three divisions when he became CEO...
-
Assume a positively sloped, short-run supply curve in Figure 19.3 and a constant $5 per unit marginal external benefi t. Show what happens with a $5 subsidy given to producers. Who gains the benefi...
-
Do the signifi cant correlations between S and per capita mortality rates prove the Social Capital and Health Hypothesis? Why or why not?
-
What is needed to find shear strain in a rock?
-
Suppose that the price of health care services rises and the quantity demanded falls. Under what conditions might the health care share of GDP fall? Rise?
-
In Figure 18.7, which depicts the utility-possibility frontier, would society ever choose an ineffi cient point (inside UU ) as the optimal point? Belinda's utility UB U UB W' min 0 UA min B W' UA U...
-
Apply effective techniques for refusing typical requests or claims and for presenting bad news to customers.
-
Suppose you are comparing just two means. Among the possible statistics you could use is the difference in means, the MAD, or the max min (the difference between the largest mean and the smallest...
-
Sketch a graph of the potential energy of two atoms as a function of the distance between them. On your graph, indicate how bond energy and bond distance are defined.
-
What is the difference between a coherent precipitate and a distinct second-phase particle?
-
What is overaging?
-
What types of heating and cooling conditions are imposed in an I-T or T-T-T diagram? Are they realistic for the processing of commercial items?
-
A company is faced with the decision of whether to lease or purchase new equipment. The equipment can be leased at an annual cost of $210,000 or purchased for $560,000. The equipment has an expected...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
A stock had returns of 28%, 1%, and 13% over the previous 3 years. The average return on this stock over the 3-year period was 14%. What was the variance of this stock's returns over the previous 3...

Study smarter with the SolutionInn App


