Use heavy and dashed wedged hues to show the shapes of the following molecules. Show the bond
Question:
Use heavy and dashed wedged hues to show the shapes of the following molecules. Show the bond dipole of each polar bond and show the overall dipole of each molecule.
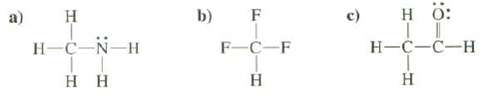
Transcribed Image Text:
a) H H-C-N-H HH b) F F-C-F H HÖ: T H-C-C-H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Use VSEPR theory to determine the geometry of the m...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the shapes of the following molecules a. CF 4 b. PCl 6 c. CO 2 d. Bh 3
-
Which of the following molecules have tetrahedral bond angles? H2O H3O+ +CH3 BF3 NH3 +NH4 -CH3
-
The ammonia molecule (NH3) has a dipole moment of 5.0 X 10-30 C m. Ammonia molecules in the gas phase are placed in a uniform electric field E with magnitude 1.6 X 106 N/c. (a) What is the change in...
-
The beam shown below is supported by a pin at A and a roller at B. The weight of the beam is 12 kN and a 15 kN force is applied 3 m to the right of A as shown. Determine the reaction forces at A and...
-
Explain why some organisations consider it important to measure customer retention and customer acquisition.
-
Airlines overbook (sell more tickets than there are seats) flights, based on past records that indicate that approximately 5% of all passengers fail to arrive on time for their flight. Sup-pose a...
-
What factors must be considered in identifying the population to be sampled?
-
Equipment replacement, no income taxes. Pro Chips is a manufacturer of prototype chips based in Dublin, Ireland. Next year, in 2010, Pro Chips expects to deliver 552 prototype chips at an average...
-
Below is a list of four problem-solving strategies created by Dawn Allison, author of Career Planning and Personal Exploration. These problem-solving strategies can be applied to any aspect of your...
-
Lets work out a few examples to get a sense of what elasticity of demand means in practice. Remember that in all of these cases, were moving along a fixed demand curveso think of supply increasing or...
-
Draw a Lewis structure for carbon monoxide (CO). Calculate the formal charges on the atoms and comment on the stability of this compound.
-
Predict the geometry at each atom, except hydrogen's, in these compounds: a) H --- 1 H b) H-c-c-c-
-
Ms. A is taxed in the top bracket and she owns a bond worth $50,000. The original cost to Ms. A was $50,000. The bond produces $5,000 of interest income. Junior is Ms. As son. He is 15 years old and...
-
Write a program to produce the shape below on CNC lathe machine. The stock is a cylinder with a diameter of 9 0 ?mm and a length of I 4 0 ?mm . ?Use two tools, the first one for roughingjand the...
-
9.19 An 8.0-in.-diameter elbow meter has a coefficient C = 0.75 when installed in a water line. The meter is connected to a mercury manom- eter having a 24-in. scale that is graduated in units of...
-
9.17 The flow of water at 150F in an existing 8.0-in.-diameter pipe line is to be measured by using a 4.0-in.-diameter ASME long-radius flow nozzle. The flow rate will vary from 0.05 to 3.0 ft/s. PAV...
-
An electrical heater is used to heat 1 litre of water from 14C to the boiling temperature at atmospheric pressure. (c) Derive an expression for the water temperature as a function of time (d) If...
-
The external loads acting on the shaft in a gear system are given as Ma = 412.5 kN mm, Tm = 366,0 kN mm, Mm = Ta = 0. For the shaft the material 1020 CD steel is selected wchic has Sut = 470 MPa....
-
Identify four corporate stakeholders and explain how they affect a businesss operations.
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
Draw the graph of the function y = 1/1 x.
-
Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the C ? C1 bonds: CI C-H Chloromethane H.
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
Make three-dimensional drawings of the following molecules, and predict whether each has a dipole moment. If you expect a dipole moment, show its direction. (a) H2C = CH2 (b) CHC13 (c) CH2C12 (d) H2C...
-
Assume you are a trader with JP Morgan. From the quote screen on your computer terminal, you notice that Bank A is quoting 0.8354/$1.00 and Bank Bis offering SF1.0913/$1.00. You learn that Bank Cis...
-
Leven Clinic uses client-visits as its measure of activity. During September, the clinic budgeted for 3,000 client-visits, but its actual level of activity was 3,050 client-visits. The clinic has...
-
If 2.25 grams of calcium hydroxide is dissolved in 154.56 ml of water, a. What's the Molarity (M), if the volume of solution is 500.00 ml? b. What's the Molarity (M) of hydroxide ions, if the volume...

Study smarter with the SolutionInn App


