Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the
Question:
Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the C ? C1 bonds:
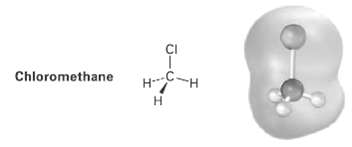
Transcribed Image Text:
CI C-H Chloromethane H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
In an electrostatic potential map the color red in...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following electrostatic potential diagrams represent H2, HCl, or NaCl. Label each, and explain your choices. (a) (b)
-
The following electrostatic potential diagrams represent CH4, NH3, or H2O. Label each, and explain your choices. a. C. b.
-
(a) Figure 11.16 is an electrostatic potential map of calicene, so named because its shape resembles a chalice (calix is the Latin word for "cup"). Both the electrostatic potential map and its...
-
A company operates a standard absorption costing system and absorbs fixed production overheads based on machine hours. The budgeted fixed production overheads for the company for the previous year...
-
Paul Barrone is a graduate student at State University. His 10-year-old son, Jamie, lives with him, and Paul is Jamie's sole support. Paul's wife died in 2015, and Paul has not remarried. Paul...
-
A hotel has 85 rooms with king beds and 125 rooms with double beds. What percentage of the hotels rooms have double beds? a. 59.5% b. 68.0% c. 85.0% d. 40.5%
-
For the HiTech bond of Example 17.4, suppose that default is recognized only at maturity and that no restitution is made, that is, the default is ignored. What is the value of that bond? - Example...
-
In year 1, AMC will earn $2000 before interest and taxes. The market expects these earnings to grow at a rate of 3% per year. The firm will make no net investments (i.e., capital expenditures will...
-
Jennifer considers a hypothetical question, and asks the group, "What if I launch the boat from the island's left end with the river current 1.5 m/s, and row at 3.0 m/s all the way to the right end,...
-
Beasanns Die-Cuts is preparing its cost of goods manufactured schedule at year-end. Beasanns accounting records show the following: The raw materials inventory account had a beginning balance of...
-
Use the electro-negatively values shown in Figure to rank the following bonds from least polar to most molar: H3C ? Li, H3C ? K, H3C ? F, H3C ? MgBr, H3C ? OH. 2.1 Li Be 1.0 1.6 Na Mg 0.9 1.2 Ca 0.8...
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
What is moral hazard and how does it relate to IMF rescue packages?
-
What are the different types of strategies organizations use to promote diversity and inclusion? Explain in the context of cultural diversity.
-
A skier starts down a 1 2 incline at 3 . 4 m / s , reaching a speed of 1 8 m / s at the bottom. Friction between the snow and her freshly waxed skis is negligible. What is the length of the incline?...
-
Discuss the VRIO framework as it relates to Aztec. How might the proposed Desron opportunity impact the current VRIO framework?
-
Ultraviolet light of wavelength 3 4 0 nm falls on a double slit. A fluorescent screen is placed 2 . 0 m away. The screen shows dark interference bands 3 . 4 cm apart. What is the distance between the...
-
The following discussion assignment will be about network operating systems. Many network operating systems are in use, and each one has strengths and weaknesses. Using the library and the Internet,...
-
Name some instances where the offense of conspiracy provides a means to protect the public from dangers incident to group activities.
-
Explain the circumstances that could result in a long-term bank loan being shown in a statement of financial position as a current liability.
-
The metal tin undergoes a transition from a gray phase to a white phase at 286 K and ambient pressure. Given that the enthalpy change of this transition is 2090 J/mol and that the volume change of...
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Show the products of these acid-base reactions and predict whether the equilibria favor the reactants or the products: a) CHCCHCCHCH + OCHCH b) CHCHNO + CHO: CH3 (c) CH3COCH, + CHCH- 10 1:Z: LL CH,...
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
What is one specific security tactics to protect the network. Identify specific tools selections to accomplish compliance and audit requirement Describe how alerts would be collected and presented...
-
1. Solve each equation for x, where 0x & a) sin x-= 0 b) cosx-3 cosx+2=0
-
Explain Article III Standing, and explain how to achieve standing. b) Explain Negligence, and provide an example of a Negligence claim. c) What happened in the case of Genesco v. Visa? d) Explain the...

Study smarter with the SolutionInn App


