Using a curved fishhook arrow, propose a mechanism for formation of the cyclopentane ring of prostaglandin H2.
Question:
Using a curved fishhook arrow, propose a mechanism for formation of the cyclopentane ring of prostaglandin H2. What kind of reaction isoccurring?
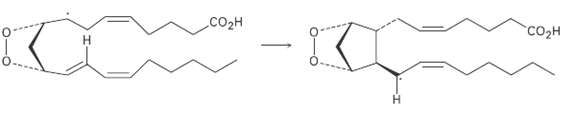
Transcribed Image Text:
CO2H "соон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Strategy The tails of the arrows show the location of ...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Write out the steps in the mechanism for formation of t-butyl alcohol in eq. 8.16. Which C-O bond cleaves, the one to the phenyl or the one to the t-butyl group? What kind of mechanism is operative...
-
Show the mechanism for formation of a Boc derivative by reaction of an amine acid with di-tert-butyl dicarbonate.
-
Write out the steps in the mechanism for formation of the product in Example 9.9. Example 9.9 Give the structure of the aldol that is obtained by treating propanal (CH3CH2CH=O) with base.
-
In the United States, a principal responsibility for preserving endangered species (e.g., a pair of endangered birds that chooses to nest on private land) and the costs of exercising that...
-
Using the table in the Financial Planning Calculations feature on page 450, complete the following table. Then answer the questions that follow the table. a. In the above situations, describe the...
-
For the nozzle described in previous problem, 4-1-44[BXF], plot how exit velocity (V2) varies with isentropic efficiency of nozzle varying from 70% to 100%, all other input parameters remaining...
-
Explain the idea behind the following heuristics: most-constrained variable most-constraining variable least-constraining variable min-conflicts
-
Your examination of the records of the Sullivan Company provides the following information for the December 31, 2007 year-end adjustments: 1. Bad debts are to be recorded at 2% of sales. Sales...
-
What are some of the issues related to sustainability and packaging? Briefly identify and explain the issues that you have identified ?
-
QUESTION 1 ?Is this statement true or false??Even if plagiarism is discovered years from now, a university can withdraw a qualification that was awarded to a student? A. True B. False 1 points ?...
-
Radical chlorination of alkanes is not generally useful because mixtures of products often result when more than one kind of CH bond is present in the substrate. Draw and name all monochloro...
-
Which of the following species is likely to be a nucleophile and which anelectrophile? (a) CH3CI (b) CH3S" (c) (d) -CH3 CH3CH
-
A partial adjusted trial balance of Frangesch Company at January 31, 2020, shows the following. Instructions Answer the following questions, assuming the year begins January 1. a. If the amount in...
-
4. A point source Gaussian plume model for a power plant uses 50 m as the effective stack height (see Figure P7.32). The night is overcast. (Note: This is not the same as "cloudy" in this model;...
-
Find the perimeter of rectangle. 10 ft 3 in. 16 ft 6 in.
-
On 1 May 2 0 1 5 , Cik Reeha, a chartered accountant with Harga Air Rumah Accountants and Partners advertised auditing services at RM 2 0 0 0 per client for a limitation period between 4 to 1 1 May 2...
-
Simplify the following expression in terms of fractional exponents. *(9x)5
-
Write the following as a single rational expression. 1 1 X X-2
-
Design a packed-bed air humidifier to process \(9.12 \mathrm{~kg} / \mathrm{h}\) of dry air. Assume that conditions are like those of Example 2.13. The packing will consist of spherical glass beads...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Construct a concept map that describes the interconnection between valence bond theory and molecular orbital theory in the description of resonance structures.
-
(a) In 1960 T. Katz (Columbia University) showed that cyclooctatetraene adds two electrons when treated with potassium metal and forms a stable, planar dianion, C8H82- (as the dipotassium salt): Use...
-
Although none of the [10]annulenes given in Section 14.7B is aromatic, the following 10 p-electron system is aromatic: What factor makes this possible?
-
Cycloheptatrienone (I) is very stable. Cyclopentadienone (II) by contrast is quite unstable and rapidly undergoes a Diels-Alder reaction with itself. (a) Propose an explanation for the different...
-
Graph the inequality. x + y -4
-
Find the slope of the line through the points (-3, 7) and (-9,2). Enter your answer as a simplified improper fraction, if necessary. Do not include "m="in your answer.
-
What is the maximum number of linearly independent vectors that can be found in the nullspace of: A = 120 3 1 24-15 4 36-1 85 4 8 1 12 8

Study smarter with the SolutionInn App


