Which of the following species is likely to be a nucleophile and which anelectrophile? (a) CH3CI (b)
Question:
Which of the following species is likely to be a nucleophile and which anelectrophile?
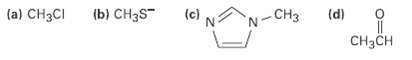
Transcribed Image Text:
(a) CH3CI (b) CH3S" (c) (d) -CH3 CH3CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
Strategy Keep in mind 1 An electrophile is electronpoor either because it is positively charged bec...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following species is not likely to have a tetrahedral shape? (a) SiBr4, (b) NF4+ (c) SF4, (d) BeCl42-, (e) BF4-, (f) AlCl4-
-
Which of the following species are tetrahedral? SiCl4, SeF4, XeF4, CI4, CdCl42?
-
Which of the following examples is likely to be caused by a somatic mutation? A. A purple flower has a small patch of white tissue. B. One child, in a family of seven, is an albino. C. One apple...
-
Water is an essential resource. For that reason moral considerations exert considerable pressure to assure that everyone has access to at least enough water to survive. Yet it appears that equity and...
-
Three years ago, you purchased 150 shares of IBM stock for $92 a share. Today, you sold your IBM stock for $183 a share. For this problem, ignore commissions that would be charged to buy and sell...
-
With a single explanatory variable, the equation used to obtain the between estimator is where the overbar represents the average over time. We can assume that E(a.) = 0 because we have included an...
-
Derive Equation 3.47. 1 Um +. (3.47) G12 Gf12 Gm
-
On what basis might Cameron maintain a suit against Anderson?
-
Coca Cola's dividend is $1/share per quarter. On average, the dividends are expected to grow by 2% per quarter. Assuming the expected return of the stock is 5% per quarter. What is the share price...
-
FOR YEAR5 sourcing has been just another word for procurement a financially material but strategically peripheral corporate function Now globalization aided by rapid technology innovation is changing...
-
Using a curved fishhook arrow, propose a mechanism for formation of the cyclopentane ring of prostaglandin H2. What kind of reaction isoccurring? CO2H "
-
An electrostatic potential map of boron trifluoride is shown. Is BF3 likely to be a nucleophile or an electrophile? Draw a Lewis structure for BF3, and explain youranswer. BF3
-
Suppose that McMaster Carr company has a monopoly in selling steel outlet boxes with knockouts. Use the following information on the demand of steel boxes to answer the following questions. a. If the...
-
Make a financial forecast for a new company like amazon for the year 2024 and 2025 Your financial forecasts should include: Cash flow statements - cash balance and the cash flow pattern Some other...
-
PROVIDE ME THE SCRIPT FOR THE FOLLOWING ROLEPLAY Simulated Activity 1 - Task 2 B: Role Play - Presentation of Communication Strategy You are now ready to present the communication strategy and...
-
When a purchaser requests the sales deposit to be invested, what special purpose Trust Account will the amount be deposited in?
-
Many birds can attain very high speeds when diving. Using radar, scientists measured the altitude of a barn swallow in a vertical dive; it dropped 1 8 8 m in 3 . 0 s . The mass of the swallow was...
-
Consider a five-year, 12% annual payment bond having a face value of 1 000 . Suppose that the bond is priced at a premium to yield 10% . Year Cash Flow PV at 10% PV/Price Yr x (PV/Pr) yr1= 120 yr 2=...
-
A key reason that influencers are so effective in marketing is that a. consumers consider influencers as more trustworthy and relatable than traditional celebrities because they are everyday people....
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
Construct a concept map that embodies the ideas of valence bond theory.
-
5-Chloro-1,3-cyclopentadiene (below) undergoes SN1 solvolysis in the presence of silver ion extremely slowly even though the chlorine is doubly allylic and allylic halides normally ionize readily...
-
Explain the following: (a) Cyclononatetraenyl anion is planar (in spite of the angle strain involved) and appears to be aromatic. (b) Although [16]annulene is not aromatic, it adds two electrons...
-
Furan possesses less aromatic character than benzene as measured by their resonance energies (96 kJ mol-1 for furan; 151 kJ mol-1 for benzene). What reaction have we studied earlier that shows that...
-
Golden Gate Mining Co. (GGMC) currently has a gold mine operating in Canada and is looking to purchase another gold mine in Peru, operating as Inca Gold Inc. (ICI). You currently work for the CFO at...
-
Cokolo Inc. has entered into the following two derivatives contracts: Purchased put options on 1,000 Trenton Co. shares that have an exercise price of $52 per share and expire in 60 days Negotiated a...
-
As a financial manager (consultant), undertake an investigation on your allocated company to evaluate its strategic financial position. Prepare a business report for the board of directors of the...

Study smarter with the SolutionInn App


