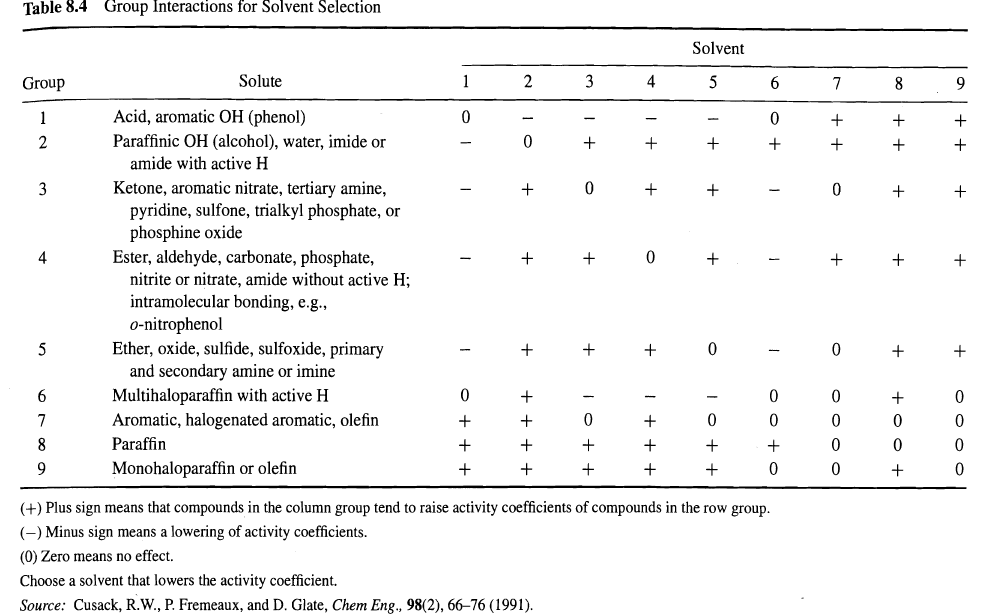
Using Table 8.4, select possible liquid liquid extraction solvents for separating the following mixtures: (a) Waterethyl alcohol,
Question:
Using Table 8.4, select possible liquid ”liquid extraction solvents for separating the following mixtures:
(a) Water”ethyl alcohol,
(b) Water”aniline, and
(c) Water”acetic acid. For each case, indicate clearly which of the two components should be the solute.
Transcribed Image Text:
Table 8.4 Solvent Solute 3 4 Group 6. Acid, aromatic OH (phenol) Paraffinic OH (alcohol), water, imide or amide with active H Ketone, aromatic nitrate, tertiary amine, pyridine, sulfone, trialkyl phosphate, or phosphine oxide Ester, aldehyde, carbonate, phosphate, nitrite or nitrate, amide without active H; intramolecular bonding, e.g., 3 4 o-nitrophenol Ether, oxide, sulfide, sulfoxide, primary and secondary amine or imine Multihaloparaffin with active H Aromatic, halogenated aromatic, olefin 6. Paraffin Monohaloparaffin or olefin (+) Plus sign means that compounds in the column group tend to raise activity coefficients of compounds in the row group. (-) Minus sign means a lowering of activity coefficients. (0) Zero means no effect. Choose a solvent that lowers the activity coefficient. Source: Cusack, R.W., P. Fremeaux, and D. Glate, Chem Eng., 98(2), 66–76 (1991).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Subject Selection of extraction solvents Given The following mixtures ...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Liquid ethyl alcohol (C2H5OH()) at 25oC is burned in a steady-flow combustion chamber with 40 percent excess air that also enters at 25oC. The products leave the combustion chamber at 600 K. Assuming...
-
The total pressure of vapor over liquid acetic acid at 71.3°C is 146 mmHg. If the density of the vapor is 0.702 g/L, what is the mole fraction of dimer in the vapor? See Problem 11.149. Problem...
-
Ethyl alcohol can be bacterially oxidized to acetic acid in the following two-step fermentation sequence: 2 C2H5OH + O2 2 CH3CHO + 2 H2O 2 CH3CHO + O2 2 CH3COOH If the alcohol-containing feedstock...
-
Midge is a 10% limited partner in Wild Catter, an oil and gas drilling partnership. She received her partnership interest in exchange for a $10,000 investment in the partnership. The balance in her...
-
A friend of your family, Nancy Chen, has just graduated from dental school and is opening up her new practice. To do this, she has formed a corporation called Chen Dental Services Ltd. Revenues from...
-
A homogeneous wire is bent into the shape shown. Determine by direct integration the x coordinate of its centroid. Express your answer in terms of a. y = kx
-
The frequency distribution from Example 7 is shown below. Find the probability of randomly selecting a social networking site user who is not 23 to 35 years old. Data from Example 7 A company is...
-
On August 10, 1978, three teenage girls died horribly in an automobile accident. Driving a 1973 Ford Pinto to their church volleyball practice in Goshen, Indiana, they were struck from behind by a...
-
Jordan Books buys books and magazines directly from publishers and distributes them to grocery stores. The wholesaler expects to purchase the following inventory: Required purchases (on account)...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
A petroleum reformate stream of 4,000 bbl/day is to be contacted with diethylene glycol to extract the aromatics from the paraflins. The ratio of solvent volume to refonnate volume is 5. It is...
-
Using Table 8.4, select possible liquid?liquid extraction solvents for removing the solute from the carrier in the following cases: Solute Carrier Ethylene glyol n-Heptane (a) Acetone (b) Ethyl...
-
Why would focusing only on the jobs currently held by people in an area tend to overstate the benefits to protectionism? Why do the numbers of those who lose jobs from eliminating trade restrictions...
-
Five identical containers (shoe boxes, paper cups, etc.) must be prepared for this problem, with contents as follows: There are five boxes containing red and white items (such as marbles, poker...
-
Find the area of a circle as a function of its circumference.
-
State whether each statement in Problems 3-12 is true or false. If it is false, explain why you think that is the case. The test point \((-2,4)\) satisfies the inequality \(y>2 x-1\).
-
Is \(\{(4,3),(5,-2),(6,3)\}\) a function? Tell why or why not.
-
Tell what the output value is for each of the function machines in Problems 15-20 for (a) 4 , (b) 6 , (c) -8 , (d) \(\frac{1}{2}\), (e) \(t\) x 421 + N/A C x +
-
Suppose that you buy an Apple iPad, you like it, and you think it will be a big seller. You expect that Apples profits will increase tremendously as a result of booming iPad sales. Should you buy...
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
A firm buys a December $100,000 Treasury bond call option with a strike price of 110. If the spot price in December is $108,000, is the option exercised?
-
It is proposed that oxygen be separated from nitrogen by absorbing and desorbing air in water. Pressures from 101.3 to 10,130 kPa and temperatures between 0 and 100 C are to be used. (a) Devise a...
-
One hundred kmol/h of a saturated-liquid mixture of 12 mol% ethyl alcohol in water is distilled continuously using open steam at 1 atm introduced directly to the bottom plate. The distillate required...
-
Figure 4.36 shows a system to cool reactor effluent and separate light gases from hydrocarbons. K-values at 500 psia and 100 F are: (a) Calculate composition and flow rate of vapor leaving the flash...
-
Show how binary exponentiation can be used to calculate a 128 efficiently. How many multiplications are required?
-
How can one tell many months the positive skew had in it?
-
Drug 4 mg I every 3 hours prn pain. Supplied as 10mg/10 ml. Calculate mL per dose?

Study smarter with the SolutionInn App


