Vapor-liquid equilibrium data at 101.3 kPa are given for the water-formic acid system. From these data, prepare
Question:
Vapor-liquid equilibrium data at 101.3 kPa are given for the water-formic acid system. From these data, prepare plots like Figures 4.7b and 4.7c. From the plots, determine the azeotropic composition and temperature at 101.3 kPa. Is the azeotrope of the minimum- or maximum-boilingtype?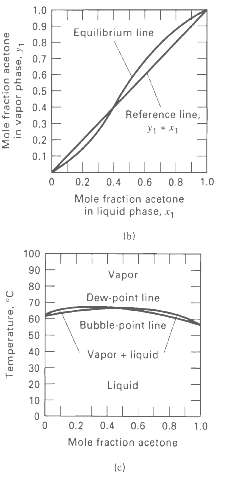
Transcribed Image Text:
1.0 0.9 Equilibrium line 0.8 0.7 0.6 0.5 D.4 Reference line, 0.3 0.2 0.1 0.2 0.4 0.6 0.8 1.0 Mole fract cn acetone in liquid phase, Ib! 100 90 Vapor 80 Dew-paint line 70 60 Bubble point line 50 40 Vapor + liquid 30 Liquid 0.2 0.4 0.6 0.8 1.0 Mole fraction acetone le) Mole fraction acetone in vapor phase, y1 Temperature, "C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
See plots below From these plots a maximumboiling a...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Vapor-liquid equilibrium data at 101.3 kPa are given for the chloroform-methanol system. From these data, prepare plots like Figures 4.6b and 4.6c. From the plots, determine the azeotropic...
-
The following equations are given for the liquid-phase activity coefficients of the water (W)-acetic acid (A) system. Find the dew point and bubble point of a mixture of composition xw = 0.5, xA =...
-
Frequency tables are given for the first 100 digits in the decimal representation of 77 and the first 100 digits in the decimal representation of 22/7. a. Construct histograms representing the...
-
The fraud examiner's report should have what characteristics? a. Accuracy and clarity b. Relevant and material information c. Should not contain opinions or prejudices of fraud examiner. d. All of...
-
Here is information for Zorba Ltd. for the year ended April 30, 2012: Total credit sales.............................................................................$2,000,000 Accounts receivable at...
-
Dubois Steel Corporation, as lessee, signed a lease agreement for equipment for five years, beginning January 31, 2017. Annual rental payments of $41,000 are to be made at the beginning of each lease...
-
What are the four specific objectives of internal accounting control?
-
Wright Company employs a computer-based data processing system for maintaining all company records. The current system was developed in stages over the past five years and has been fully operational...
-
Records available for the latest period are as follows: Support Depts. Customer Service Administration 580,000 $ 7 100 Total Costs $ # of Employees # of Customer Inquiries Operating Depts. Shirts...
-
Maury bought one full bitcoin in March 2021. His basis in the coin is $6,118. In July of 2022, he donated 0.2 of the coin to a qualified charitable organization. The fair market value of a full coin...
-
Using vapor pressure data from Exercises 4.6 and 4.8 and the enthalpy data provided below: (a) Construct an h-x-y diagram for the benzene-toluene system at 1 atm (101.3 kPa) based on the use of...
-
Vapor-liquid equilibrium data for mixtures of water and isopropanol at 1 atm (101.3 kPa, 760 torr) are given below. (a) Prepare T-x-y and x-y diagrams. (b) When a solution containing 40 mol %...
-
In Exercises 112, write the first four terms of each sequence whose general term is given. a n = (-1) n+1 (n + 4)
-
State whether the rate of change for the more recent 5-year period (20072011) was lower than or higher than the rate of change for the earlier 5-year period (20022006) in each country: (a) the United...
-
Determine the output of the Hilbert transformer designed in Exercise 5.23 to the input signal \(\mathrm{x}\) determined as Fs \(=1500 ; \mathrm{TS}=1 / \mathrm{Fs} ; \mathrm{t}=0: \mathrm{TS}:...
-
Use the NPV method to determine whether Farran Company should invest in the following projects: Project A: Costs $250,000 and offers six annual net cash inflows of $75,000. Farran Company requires...
-
For the fiscal year ended 31 December 2012, WPP Group PLC (London: WPP) reported basic EPS of 66.2 and diluted EPS of 62.8. Based on a closing stock price of 1058.0 on 1 February 2013, the day on...
-
Design a bandpass filter satisfying the specification below using the WLS and Chebyshev methods. Discuss the trade-off between the stopband minimum attenuation and total stopband energy when using...
-
With regard to gene duplications, which of the following statement(s) is/are correct? a. Gene duplications may be caused by nonallelic homologous recombination. b. Large gene duplications are more...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Discuss the most popular forms of ETF investment strategies.
-
A mixture of propionic and n-butyric acids, which can be assumed to form ideal solutions, is to be separated by distillation into a distillate containing 95 mol% propionic acid and a bottoms of 98...
-
Rigorous, computer-based methods for multicomponent distillation are readily available in process simulators. Why, then, is the FUG method still useful and widely applied for distillation?
-
The enzyme alcohol dehydrogenase (ADH) catalyzes oxidation of primary and secondary alcohols to aldehydes and ketones. It can be produced from a culture of Lactobacillus brevis, and recovered from...
-
9 1. Compute the indefinite integral. +1 (3+ 4+4) dr.
-
Sarah Jackson Antiques issued its 11%, 10-year bonds payable at a price of $338,220 (face value is $400,000). The company uses the straight-line amortization method for the bond discount or premium....
-
Suppose you start with a full tank of gas (14 gallons) in your truck. After driving 6 hours, you now have 6 gallons left. If x is the number of hours you have been driving, then y is the number of...

Study smarter with the SolutionInn App


