We?ve seen that the Diels?Alder cyclo addition reaction is a one-step, pericyclic process that occurs through a
Question:
We?ve seen that the Diels?Alder cyclo addition reaction is a one-step, pericyclic process that occurs through a cyclic transition state. Propose a mechanism for the following reaction:
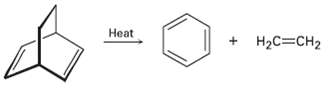
Transcribed Image Text:
Нeat Нeat + H2C=CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
DielsAlder reaction are reversible when the produ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. Electron flow arrows should start on the...
-
The following list shows the top six pharmaceutical companies in the United States and their sales figures ($ millions) for a recent year. Use this information to construct a pie chart and a bar...
-
What are the pros and cons of offering sick leave benefit?
-
Consider a reaction A2(g) + B2(g) 2 AB(g), with atoms of A shown in red in the diagram and atoms of B shown in blue. (a) If Kc = 1, which box represents the system at equilibrium? (b) What is the...
-
Partly to address the existence and completeness assertions, auditors typically test purchase cutoff at the balance sheet date, thereby addressing whether purchase transactions are recorded in the...
-
A shirt manufacturer buys cloth by the 100- yard roll from a supplier. For setting up a control chart to manage the irregularities (e. g., loose threads and tears), the following data were collected...
-
How is performance management at dmg world media different from traditional performance management systems at other companies? What was the impetus for developing a different performance management...
-
Problem #12: Planning and Control Don't-Cha-Wanna-a-Coffee, a large-scale coffee company that has a chain of 125 retail coffee stores throughout New England, was embroiled in an ethics scandal 3...
-
Norbornadiene (Problem 14.41) can be prepared by reaction of chloroethylene with 1, 3-cyclopentadiene, followed by treatment of the product with sodium ethoxide. Write the overall scheme, and...
-
In light of your answer to Problem 14.43, propose a mechanism for the followingreaction: CH CH + CO2 Heat "CCH do,c, H a-Pyrone CHC-
-
Explain why a DNA polymerase that could synthesize DNA in the 3' 5' direction would have a selective disadvantage even if it had 5' 3' proofreading activity.
-
Discuss the concept of demand paging in the context of virtual memory systems. How does it optimize memory usage by selectively loading portions of a process into physical memory based on demand and...
-
On January 1, 2017, a company began constructing a new asset. It borrowed $1,000,000 to finance the construction with an annual interest rate of 5%. Assume the following payments for construction...
-
Use the following information (in random order) from a merchandising company and from a service company. McNeil Merchandising Company Accumulated depreciation Beginning inventory Ending inventory...
-
Bond Corporation is an electing S corporation with two equal shareholders (A and B). In 2022 Bond had separately computed income of 100,000 and a non-separately computed loss of $500,000. Before...
-
Flowers Ltd. is a Canadian business operating in the outdoor furniture industry. The company manufactures and distributes outdoor furniture to Canadian retailers. You work in the accounting...
-
If a \({ }_{92}^{238} \mathrm{U}\) nucleus is struck by a neutron, it may absorb the neutron. The resulting nucleus then rapidly undergoes betaminus decay. The daughter nucleus of that decay is A....
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
A solution contains 3.95 g of carbon disulfide (CS 2 ) and 2.43 g of acetone (CH 3 COCH 3 ). At 35 C the vapor pressures of pure carbon disulfide and pure acetone are 515 torr and 332 torr,...
-
Predict the major products of the following reactions: (a) (b) (c) (d) Zn(Hg) HCI, H20 (1) H2NNH (2) KOH, heat (1) N2H4 2) KOH, heat O HCI, H,O
-
Use equations to show the fragmentation leading to each numbered peak in the mass spectrum of octan-2-one. 100 80 e 60 2 40 20 0 43 85 113 12 10 20 30 40 50 60 70 80 90 100 110 120 130 40 150 160
-
The following compounds undergo McLafferty rearrangement in the mass spectrometer. Predict the masses of the resulting charged fragments. (a) Pentanal (b) 3-methylhexan-2-one (c) 4-methylhexan-2-one
-
Given vectors, v = (1, 1) and w = (5, 4), find the following. v+w= ||vw|| = ||v|| + |||| =
-
If a bank savings account with $ 2 , 4 1 0 will be worth $ 3 , 9 2 1 in 2 years, and if the interest rate is constant, what is the interest rate quote that the bank would post in its window?
-
Solve x2-24x+14= 0

Study smarter with the SolutionInn App


