What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all
Question:
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities.
(a) 2-Methyl-2-propanol
(b) 1-Ethylcyclohexanol
(c) 3-Phenyl-3-pentanol
(d)2-Phenyl-2-pentanol
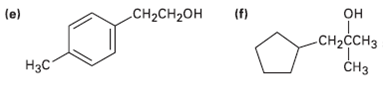
Transcribed Image Text:
(f) CH-CH2он (e) он CH2CCH3 Нэс CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
Grignard Reagent Carbonyl Compound a b CH3MgBr 2 CHMgBr or CH3CHMg Br i CH3CCH...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkynes would you start with to prepare the followingketones? (a) (b) CHCHH2H CH3CH2CH2CH3
-
What alkyne would you start with to prepare each of the following compounds by a hydroboration/oxidationreaction? (b) (a) CH CH-CCHCH3 -CH2CH CH
-
What ester an what Grignard reagent might you start with to prepare the following alcohols? (b) , H (c) HCH-H2H2,H CH-CH (a)
-
Oliveras Company had net credit sales during the year of $800,000 and cost of goods sold of $500,000. The balance in accounts receivable at the beginning of the year was $100,000, and the end of the...
-
Describe the differences between multinational and global firms.
-
Let a > 0. Show that the series (1 + an)-1 is divergent if 0 < a < 1 and is convergent if a > 1.
-
What classes of transactions are included in the expenditure cycle?
-
Seabreeze Furniture in Orlando maintains a large central warehouse where it stores items until they are sold or needed by the companys many stores in the central Florida area. A four-person crew...
-
How would you define "art"? What makes something a work of art? What are the characteristics of a work of art-according to you, not according to the textbook, a dictionary, or a website? In this...
-
Srf is but an approximation of prf Explain.
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Evidence for the intermediate carbocations in the acid-catalyzed dehydration of alcohols comes from the observation that rearrangements sometimes occur. Propose a mechanism to account for the...
-
Use the Taylor series in Table 9.5 to find the first four nonzero terms of the Taylor series for the following functions centered at 0. ln (1 + x 2 )
-
What is self paced interview ? What kind of questions come in this kind of interviews?
-
A farmer grows wheat, which he sells to a miller for $3,000. The miller produces flour , which he sells to a baker for $3,600. The baker produces all sorts of bread , which are sold to the public for...
-
For the past 50 years, median housing prices have grown on average 5.4% per year. The Baltimore Neighborhood Indicators Alliance gives the median price of homes sold for the following Baltimore City...
-
Emilia (age 24) places 2,535,000 in trust with income to Andy (age 32) and Victoria (age 38) for 35 years, remainder to Jill (age 18). Emilia does not reserve any powers to alter, amend, revoke or...
-
Provide an explanation about how you determine the project status based on the data? Provide an explanation about how process, project, and product metrics each explain a different part of your...
-
The cost of goods sold computations for Gouda Company and Edam Company are shown below. Instructions a. Compute inventory turnover and days in inventory for each company. (Round inventory turnover to...
-
An annual report of The Campbell Soup Company reported on its income statement $2.4 million as equity in earnings of affiliates. Journalize the entry that Campbell would have made to record this...
-
In a 1999 study of cobalt nanocrystals, D. P. Dinega and M. G. Bawendi discovered that cobalt forms an interesting cubic structure unlike any of the cubic structures described in this chapter. They...
-
Fischer projection formulas are often used to depict compounds such as glyceraldehyde, lactic acid, and tartaric acid. Draw Fischer projections for both enantiomers of (a) Glyceraldehyde, (b)...
-
Designate the (R) or (S) configuration at each chirality center in the following molecules. Cl SH Br Br
-
Albuterol, shown here, is a commonly prescribed asthma medication. For either enantiomer of albuterol, draw a three-dimensional formula using dashes and wedges for bonds that are not in the plane of...
-
Consider a 15%, 20 year bond that pays interest annually, and its current price is $850. What is the promised yield to maturity?
-
You are a dual-income, no-kids family. You and your spouse havethe following debts (total): mortgage, $212,000; auto loan,$22,000; credit card balance, $16,000; other debts, $22,000.Further, you...
-
Can you please provide a feedback to this post? 1. What is the managerial view on business? What are the stakeholders view on business? How do they differ? The management viewpoint on business...

Study smarter with the SolutionInn App


