What alkynes would you start with to prepare the followingketones? (a) (b) CHCHH2H CH3CH2CH2CH3
Question:
What alkynes would you start with to prepare the followingketones?
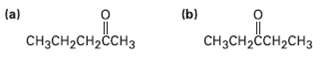
Transcribed Image Text:
(a) (b) CHзCHассH2сHз CH3CH2CH2ČCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
a b CH3CHCHCCH H3O HgSO4 OH CH3CH...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What alkenes would you start with to prepare the following alkylhalides? CH-CH (a) (b) Br Br CI (c) (d) CH3CH2CHCH2CH2CH3
-
What alkyne would you start with to prepare each of the following compounds by a hydroboration/oxidationreaction? (b) (a) CH CH-CCHCH3 -CH2CH CH
-
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities. (a) 2-Methyl-2-propanol (b) 1-Ethylcyclohexanol (c) 3-Phenyl-3-pentanol...
-
Ancient Indians did not believe in war and violence when it came it an expansion of political territory but relied on peaceful negotiation and strategy. Please explain your views on this quote with...
-
A computer manufacturer sells its laptop model through a web-based distributor, who buys at a unit cost of $200 and sells at a unit price of $500. The product life cycle is so short that the...
-
A element of material in plane strain (see figure) is subjected to strains, (x = 480 ( 10-6, (y = 70 ( 10-6, and (xy = 420 ( 10-6. Determine the following quantities: (a) the strains for an element...
-
Why is it sometimes difficult to differentiate between types of social media? For example, Twitter is often referred to as an SNS, a microblog, and a social bookmarking service. YouTube is often...
-
The trial balance as of July 31, 2017, for Sandra Sousa, Registered Dietician, is presented below: Requirements 1. Prepare the income statement for the month ended July 31, 2017. 2. Prepare the...
-
Explain the skills required for effective grievance and discipline-handling procedures.
-
Sullivans Island Company began operating a subsidiary in a foreign country on January 1, 2020, by investing capital in the amount of 60,000 pounds. The subsidiary immediately borrowed 140,000 pounds...
-
What product would you obtain by hydration of the following alkynes? (a) CH3CH2CH2C=CCH2CH2CH3 CH (b) CCH2CECH-CH2CH3
-
How would you prepare the following carbonyl compounds starting from an alkyne (reddish brown ? Br)? (b) (a)
-
Consider the following information: 1. On December 1, 2005, a U.S. firm plans to sell a piece of equipment [with an asking price of 200,000 units of a foreign currency (FC)] during January of 2006....
-
What are sales and use taxes? How do they affect a firm's liabilities? working for a company which buys parts and material for construction projects, what part does the company play in paying or...
-
The following data are for Lauren Enterprises: Selling and administrative expenses Direct materials used Direct labor (25,000 hours) Factory overhead application rate $75,000 $265,000 $300,000 $16...
-
Variable manufacturing costs at ACME Corp. are as follows: Direct materials $3.25 per unit Direct labor $1.05 per unit Fixed manufacturing costs are $6,000 per month. If the company produces 4,500...
-
Ying's Mount Robson Guide Tours business is headquartered out of British Columbia and operates only during the summer season. This year Ying grossed $38,656 including GST and PST. She also spent...
-
A 110 g mass on a 1.7-m-long string is pulled 7.8 to one side and released Part A How long does it take for the pendulum to reach 2.6 on the opposite side? Express your answer with the appropriate...
-
When solving physics problems, what are the advantages of making simplified visual representations of the situations?
-
The swap spread is the difference between the swap rate and the equivalent-maturity Treasury bond yield. Explain why a widening swap spread may be a signal of deteriorating economic conditions. Plot...
-
A 25.00-mL sample of an unknown HClO 4 solution requires titration with 22.62 mL of 0.2000 M NaOH to reach the equivalence point. What is the concentration of the unknown HClO 4 solution? The...
-
(a) In most peptides, the amide bonds have the Z conformation; explain why. (b) One particular amino acid residue in the PepC position adopts the E conformation in some cases. Which amino acid...
-
When N - acetyl- L -aspartic acid is treated with acetic anhydride, an optically active compound A, C6H7NO4, is formed" Tleaffient of A with the amino acid L -alanine yields two separable, isomeric...
-
When N - acetyl- L -aspartic acid is treated with acetic anhydride, an optically active compound A, C6H7NO4, is formed" Tleaffient of A with the amino acid L -alanine yields two separable, isomeric...
-
1. What is difference between Torque and Force? Explain with one example of each. 2. What is difference between linear momentum and angular momentum? Explain with one example of each. 3(a) What is...
-
On August 30, JumpStart paid the following expenses: August rent, $2,300 August's utility bill, $525 Employee wages, $1,750 Parking lot cleaning fee, $27 Journalize these payments as one journal...
-
A 700 g ball strikes a wall at 15.1 m/s and rebounds at 14.4 m/s. The ball is in contact with the wall for 0.034 s. What is the magnitude of the average force acting on the ball during the collision?...

Study smarter with the SolutionInn App


