What fragments might you expect in the mass spectra of the followingcompounds? (c) (b)
Question:
What fragments might you expect in the mass spectra of the followingcompounds?
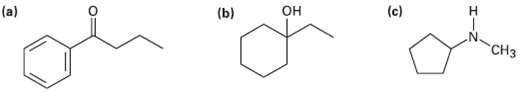
Transcribed Image Text:
(c) (b) он н "СНз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
a This ketone shows mass spectrum fragments that are due to alpha cleavage and ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown in Figure 13.7. Match the compounds with the spectra. 100 73 80 S 60 57 20 0 10 20 30 40 50 60 70 80 90...
-
What kind of returns might you expect in the stock market? One way to measure how the stock market has performed is to examine the rate of return of the S&P 500 Index. To see historical prices of the...
-
What changes would you expect in the standard deviation for a portfolio of between 4 and 10 stocks, between 10 and 20 stocks, and between 50 and 100 stocks?
-
Shock Electronics sells portable heaters for $35 per unit, and the variable cost to produce them is $22. Mr. Amps estimates that the fixed costs are $97,500. a. Compute the break-even point in units....
-
It has been said that Porter's five-force analysis turns antitrust law on its head. What do you think this means?
-
Dichlorobenzene, C6H4Cl2, exists in three forms (isomers) called ortho, meta, and para: Which of these has a nonzero dipole moment? Explain. Cl Cl Cl Cl CI Cl ortho meta para
-
Why is it important that a company provide its employees with a secure and respectful workplace?
-
Carmen Camry operates a consulting firm called Help Today. On August 31, the companys records show the following accounts and amounts for the month of August. Use this information to prepare an...
-
The Company provides interpretation services to many public sectors, including police, hospitals, schools, provincial workers' compensation boards, and the social services department of the federal...
-
Loan payments of $3000 due one year ago and $2500 due in four years are to be replaced by two equal payments. The first replacement payment is due now and the second payment is due in six years....
-
Assume that you are in a laboratory carrying out the catalytic hydrogenation of cyclohexane to cyclohexane. How could you use a mass spectrometer to determine when the reaction is finished?
-
How might you use IR spectroscopy to distinguish among the three isomers 1 -butyne, 1, 3-hutadiene, arid 2-butyne?
-
On April 30, 2019. one year before maturity. Weber Company retired $200.000 of 9% bonds payable at 101. The book value of the bonds on April 30 was $197,600. Bond interest was last paid on April 30 ,...
-
John purchases State of Oklahoma general-purpose bonds at a cost of $3,400 in 2018. He receives $210 interest on the bonds in 2018 2019, and 2020. In 2020, he sells the bonds for $3,800. How much...
-
Solve for x. 25x-7= 8 x =
-
Evaluate 3x-9 when x = -2. The value is
-
: What is the difference between containerization and virtualization in the context of operating systems? Discuss the benefits and limitations of each approach with respect to resource management,...
-
(a) If log 9 = 2, then x = (b) If log 16 = 2, then =
-
A stunt performer falls from the roof of a two-story building onto a mattress on the ground. The mattress compresses, bringing the performer to rest without his getting hurt. Is the work done by the...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
Find the mass of urea (CH 4 N 2 O) needed to prepare 50.0 g of a solution in water in which the mole fraction of urea is 0.0770.
-
Predict the products of the following reactions. ether hexane + 2 Li hexane + 2 Li
-
Show how you would synthesize the following primary alcohols by adding an appropriate Grignard reagent to formaldehyde. (a) (b) (c) CH,OH CH,OH
-
Show two ways you could synthesize each of the following secondary alcohols by adding an appropriate Grignard reagent to an aldehyde. (a) (b) (c)
-
1. Define human resource (HR) management and explain how it relates to the management process. Cite examples of the application of these concepts, preferably from personal, professional experience.
-
Mickley Corporation produces two products, Alpha6s and Zeta7s, which pass through two operations, Sintering and Finishing. Each of the products uses two raw materialsX442 and Y661. The company uses a...
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...

Study smarter with the SolutionInn App


