What is the hybridization at all atoms, except hydrogen's in these compounds? a) CHNH, d) b) CH=CHCHC=N
Question:
What is the hybridization at all atoms, except hydrogen's in these compounds?
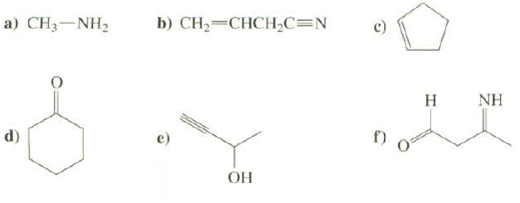
Transcribed Image Text:
a) CHạ—NH, d) b) CH₂=CHCH₂C=N OH 6 H NH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a CH3NH2 sp sp d Sp3 sp3 O s...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
What is the hybridization at the indicated atoms in these compounds? a) CHCH=CH_NHCH3 12 3 c) CH,=CH0CH, 1 2 3 4 5 0: b) CHC0CH, 1 d) 2 1 NH
-
What is the hybridization of the indicated atom in each of the following molecules? a. b. c. d. e. f. CH CH CH CH CCH CH3CH2OH CH3CH NCH3 CH:OCH CH
-
y Working this on the www.bis.doc.gov website. "MY company makes fingerprinting powder kits for both domestic and external clients. It is a popular item in my inventory. I have a new customer in...
-
Explain why some organizations might prefer to use variable cost rather than absorption cost as a basis for setting transfer prices?
-
Jaycees department store chain is planning to open a new store. It needs to decide how to allocate the 100,000 square feet of available floor space among seven departments. Data on expected...
-
The following data are taken from measurements on Indian Fork Creek: \(A=26 \mathrm{~m}^{2}, P=16 \mathrm{~m}\), and \(S_{0}=0.02 \mathrm{~m} / 62 \mathrm{~m}\). Determine the average shear stress on...
-
The general ledger of Red Storm Cleaners at January 1, 2021, includes the following account balances: The following is a summary of the transactions for the year:1. March 12 Provide services to...
-
=== Let r(t) (2,1 t, 4t). Calculate the derivative of r(t) a(t) at t = 8, assuming that a(8)=(-3, -9, 6) and a'(8)=(-2,-2,3) d. dt (t) - a(t)|t-s=
-
Every college student had the problem of selecting the college or university to attend. Was this a simple, intermediate, or complex problem for you? Explain.
-
Show the location of the two planar nodes in this 3d atomic orbital?
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it? (both) TTT a) HC=C_C_CH ...
-
What is the key question distinguishing relevant data from irrelevant data?
-
Discuss why, in a relational database, first normal form is necessary. Is it sufficient to reduce redundancy? Why?
-
Particle A, located at the origin, carries a charge of \(6.0 \mathrm{nC}\). (a) What is the electrostatic potential (relative to zero potential at infinity) at a position \(r=8.0 \mathrm{~cm}\) from...
-
An electron and a proton are fixed at the vertices of an equilateral triangle with sides of length \(a\). (a) What is the potential (relative to zero at infinity) at the apex of the triangle. \((b)\)...
-
Using information from the Application "Flight Insurance," calculate the price of fair insurance if the probability were as high as 0.00000077 , the frequency in 2001 when many people died in the...
-
(a) What is the potential at a point \(50 \mathrm{~cm}\) away from a point charge with a magnitude of \(3.0 \mathrm{nC}\) ? (b) How would the potential change if you measure the potential at a point...
-
In Sec. 3.5 we discussed various aspects of relations between dynamics at different levels of organisation in a complex system. (a) Think of the human body and try to list the different time scales...
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
Is the series convergent or divergent? 22"31-n n=1
-
Draw and name all monochloro derivatives of 2, 5-dimethylhexane, C8H17C1.
-
Predict the hybridization of the carbon atom in each of the following functional groups: (a) Ketone (b) Nitrile (c) Carboxylic acid
-
Draw the structures of the following molecules: (a) Biacetyl, C4H6O2, a substance with the aroma of butter; it contains no rings or carboncarbon multiple bonds. (b) Ethylenimine, C2H5N, a substance...
-
The bank reconciliation for Widgets Inc. included the following items: $30 bank service charge; NSF check of $275; bank error of $30; EFT collection from a customer for $2,335. Based on the...
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
A simply supported girder bridge is to support vehicular loads. It is anticipated that the worst load will come from a three-axle truck shown in Figure I below. Assuming the self-weight of the girder...

Study smarter with the SolutionInn App


