Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of
Question:
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it?
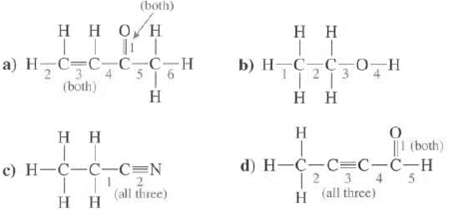
Transcribed Image Text:
(both) Η Η Ο Η TTT a) HC=C_C_CH Το (both) H Η Η 0 H-C-C-C=N 11 Η Η 2 (all three) Η Η || b) H_C_CO Η Η Η H |1 (both) d) H=C=C=C-C-H 1 2 3 4 H (all three)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (18 reviews)
a 1 3 5 Csp2Osp2 and TTC2p02p 2 OCsp2H1s Csp2Csp2 and TTC2pCp2 4 OCsp2Csp2 Csp2...View the full answer

Answered By

Loise Ndungu
I have five years of experience as a writer. As I embark on writing your papers from the prologue to the epilogue, my enthusiasm is driven by the importance of producing a quality product. I put premium product delivery as my top priority, as this is what my clients are seeking and what makes me different from other writers. My goal is to craft a masterpiece each time I embark on a freelance work task! I'm a freelance writer who provides his customers with outstanding and remarkable custom writings on various subjects. Let's work together for perfect grades.
4.90+
78+ Reviews
224+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
Indicate the type of atomic orbitals that are overlapping to form each of the different kinds of bonds in CH3OCH3 (For example, a carbon sp3 AO and a hydrogen Is AO). What kinds of orbitals are...
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
The general term that refers to the tendency of a parcel of air to either remain in place or change its initial position is ________. a. adiabatic b. conditional instability c. stasis d. stability
-
Explain why it may not be desirable for head office management to dictate transfer prices. Can you provide an example of a situation where this may be appropriate?
-
Floyd's Bumpers pays a transportation company to ship its product in full truckloads to its customers. Therefore, the cost for shipping is a function of the distance traveled and a fuel surcharge...
-
What is a CDR and what is it intended to accomplish?
-
Presented below are transactions related to Sayid Company. 1. On December 3, Sayid Company sold $570,000 of merchandise to Shephard Co., terms 2/10, n/30, FOB shipping point. The cost of the...
-
A. Opal Ltd. has just commissioned the construction of a new factory at an agreed cost of 6m. Work started on 1 April 2022 and was completed and opened for business on 31 December 2022, the last day...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
What is the hybridization at all atoms, except hydrogen's in these compounds? a) CHNH, d) b) CH=CHCHC=N OH 6 H NH
-
What is the hybridization at all atoms, except hydrogen's, in these compounds? a) H H H H H H H H CH 4.6 c) HC-N-CH3 : : g) CHC-OH h) CHC-NHCH,
-
Volkswagen obfuscated, they denied, and they ultimately lied. These were the words of U.S. Attorney General Loretta Lynch, as she set out how the German carmaker would be punished for attempting to...
-
To solve the separable differential equation we must find two separate integrals: dy = help (formulas) and 10 da = help (formulas) The first integral we integrate by substitution: help (formulas)...
-
At John Deere, thousands of people both inside and outside the organization make decisions about target segments, branding, product development, pricing, promotion, and distribution. They talk with...
-
Let S be the set that contains the smallest 100 positive integers; that is, S = {1, 2, 3, ... , 99, 100}. Prove that a list can be made of the 2100 subsets of S so that the empty set is the first...
-
a u au at x 0 0, u(x, 0) = 0, h=1/12 If then U,1 = O = O U,1 = U,1 = 1/2 (U+ 10 + U4-10) + 1/1 4 O Ui, 1 subjected to the conditions (x, 0) = 1 2 (U+1,1 + U-1,1)+ = 1/1 (14+10 + 24-10) i+ 2 at 1 U,1...
-
Consider the following statement in set notation. Give an explanation in words of what the statement is saying, and state whether it is a true or false statement, giving justification. X 3x EZ such...
-
After rereading the Opening Profile, identify all effective actions Ellie Symes and Wyatt Wells initiated in the early days of building The Bee Corp.
-
Trade credit from suppliers is a very costly source of funds when discounts are lost. Explain why many firms rely on this source of funds to finance their temporary working capital.
-
Prove that e x is equal to the sum of its Maclaurin series.
-
Draw structures for the following: (a) 2-Methyiheptane (b) 4-Ethyl-2, 2-dimethylhexane (c) 4-Ethyl-3, 4-dimethyloctane (d) 2, 4, 4-Trimethylheptane (e) 3, 3-Diethyl-2, 5-dimethylnonane (f)...
-
Draw a compound that: (a) Has only primary and tertiary carbons (b) Has no secondary or tertiary carbons (c) Has four secondary carbons
-
Draw a compound that: (a) Has nine primary hydrogens (b) Has only primary hydrogens
-
The recent collapse of two major banks, Silicon Valley Bank and Signature Bank, and the subsequent federal intervention to protect uninsured depositors, has reignited debates about government...
-
Privatizing Social Security in the United States has been a topic of debate for many years, with strong opinions on both sides since its creation as part of President Franklin D. Roosevelt's New...
-
Economists measure the price level by using a basket of goods and services and calculating how the total cost of buying that basket of goods will increase over time. Economists often express the...

Study smarter with the SolutionInn App


