What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H.
Question:
What kind of hydribization do you expect for each carbon atom in the followingmolecules?
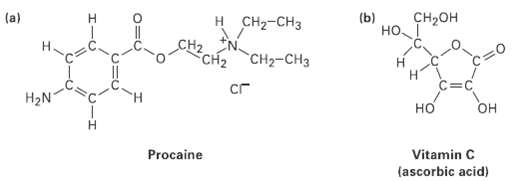
CH-он (b) H. н Cна-сHз (a) но 1 "CH-CHз CH2 C=C сг H2N H. но он Procaine Vitamin C (ascorbic acid)
Step by Step Answer:
a HN St H 302 H sp3 sp CC2 H ...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Organic Chemistry questions
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What dividends do you expect for Goodman Industries stock over the next 3 years if you expect the dividend to grow at the rate of 5% per year for the next 3 years? In other words, calculate D1, D2,...
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
Discuss the ways that managed care organizations can infl uence the adoption of new technologies.
-
Review Examples 4 and 6 in the text. In both examples, the taxpayer's AGI is $129,400 even though in Example 6 there is $700 of nonrecaptured 1231 loss from 2015. Explain why the two AGI amounts are...
-
The U.S. Federal Trade Commission assesses fines and other penalties against weight-loss clinics that make unsupported or misleading claims about the effectiveness of their programs. Brochures from...
-
a. What is the homeowner's daily energy requirement as measured from the load? b. If she replaces her alarm clock with a wind-up clock, how much energy per day will she avoid using? c. What would you...
-
The stockholders equity accounts of Ashley Corporation on January 1, 2012, were as follows. Preferred Stock (8%, $50 par, cumulative, 10,000 shares authorized)..... $ 400,000 Common Stock ($1 stated...
-
Why do you need to calculate merchandise handled and EOM stock for the season to cost ? describe please.
-
Oasis Health Care Inc. is owned and operated by Dr. George Hancock, the sole stockholder. During January 2013, Oasis Health Care entered into the following transactions: Jan. 1 Received $15,000 from...
-
Potassium methoxide, KOCH3, contains both covalent and ionic bonds. Which do you think is which?
-
Pyridoxal phosphate, a close relative of vitamin B6, is involved in a large number of metabolic reactions. Tell the hydribization, and predict the bond angles for each non-terminalatom. . Pyridoxal...
-
Find the following derivatives. All letters stand for constants except for the dependent and independent variables indicated: a. dy/dx, where y = (ax 2 + bx + c) 3/2 . b. d ln (P)/dT, where P = ke...
-
A car sells for 7 5 0 0 originally and depreciates in value by 1 8 % each year for 1 5 years. at this time the car, still in mint condition becomes a classic and begins to increase in value by 2 5 %...
-
Find the center and radius if the circle with the given equations. a ) x 2 + y 2 = 9
-
Find the quadratic polynomial whose graph goes through the points ( 1 , 4 ) , ( 0 , 4 ) , and ( 1 , 1 2 ) .
-
Multiply. (-32) (-17)
-
Solve: ex2+6 - = 65
-
Can every histogram be converted to a three-dimensional graphic? Does the three-dimensional version of a histogram have more visual appeal? Does the three-dimensional version of a histogram provide...
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure and fugacity of nitrogen as a function of specific volume at the two temperatures a. 110 K b. 150 K
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
Ethics in Human Resource Practices HR teams will play vital roles in organizations to ensure ethical behavior and compliance. To be effective, they must first place an emphasis on ethical behavior...
-
Why do managers struggle with ethics when it comes to making strategic management decisions for their organization?
-
Discuss the elements of effective teamwork presented by each: In what ways are they alike? In what ways are they different? Discuss whether a high performing team requires a team leader. Why or why...

Study smarter with the SolutionInn App


