What product would you obtain from catalytic hydrogenation of the following alkenes? CH2H (a) (b) CH CH
Question:
What product would you obtain from catalytic hydrogenation of the following alkenes?
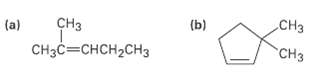
Transcribed Image Text:
сНз Cнзс—снсH2сHз (a) (b) CHз "CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
Catalytic hydrogenation produces alkanes fr...View the full answer

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What Product would you obtain from a base-catalyzed Michael reaction of 3-button-2-one with each of the following nucleophilicdonors? (b) o (a) ELOCH,OET .Et
-
What product would you obtain from a base-catalyzed Michael reaction of 2, 4-pentanedione with each of the following , -unsaturated acceptors? (a) 2-Cyclohexenone (b) Propene nitrile (c) Ethyl...
-
What product would you expect to obtain from catalytic hydrogenation of natural rubber? Would the product be syndiotactic, atactic, or isotactic?
-
Consider the deletion of record 5 from the file as shown below compare the relative merits of the following techniques for implementing the deletion: a. Move record 6 to the space occupied by record...
-
The Fischer Theatre compared attendance at its Saturday and Sunday matinee performances of a major Broadway musical. At = .05, is the Sunday matinee attendance significantly greater than the...
-
Consider a spring with k = 0.1 and m = 1.0. Find the period T that produces a solution of the spring equation. Is this spring stronger or weaker than one with k = 1.0, and does the oscillation have a...
-
Discuss the techniques of value planning, value engineering, and value analysis.
-
Bank Reconciliation and Adjusting Entries Aragon Company has just received the August 31, 2010, bank statement, which is summarized below. The general ledger Cash account contained the following...
-
3. Consider the following class definition and write an appropriate Java program according to the definition and output the result. public class Children [String name; int friends; public...
-
Martha's Vineyard Marine Supply is a wholesaler for a large variety of boating and fishing equipment. The company's controller, Mathew Knight, has recently completed a cost study of the firm's...
-
What products would you expect from the following reactions? CH2 (a) + CHCI3 CH (b) Zn(Cu) CH2I2 H2HH
-
What product would you expect from reaction of cis-2-butene with meta-chloro-peroxybenzoic acid? Show the stereo chemistry.
-
Sketch the graph, noting the transition points and asymptotic behavior. y = X (x - 4)2/3
-
heridan Manufacturing has equipment that cost $ 7 5 2 4 0 0 and has accumulated depreciation of $ 3 4 2 0 0 0 . When the equipment has a fair value of $ 6 8 4 0 0 0 , it is exchanged for equipment...
-
Hudson Company reports the following contribution margin income statement. HUDSON COMPANY Contribution Margin Income Statement For Year Ended December 31 Sales (11,000 units at $300 each) Variable...
-
Lansing Company s current - year income statement and selected balance sheet data at December 3 1 of the current and prior years follow. LANSING COMPANY Income Statement For Current Year Ended...
-
Fickel Company has two manufacturing departments-Assembly and Testing & Packaging. The predetermined overhead rates in Assembly and Testing & Packaging are $16.00 per direct labor-hour and $12.00 per...
-
A company applies overhead at a rate of 1 5 5 % of direct labor cost. Actual overhead cost for the current period is $ 8 2 5 , 0 0 0 , and direct labor cost is $ 5 0 0 , 0 0 0 . Determine whether...
-
What elements make up 98, of the Earth by weight?
-
Apply Jacobis method to the given system. Take the zero vector as the initial approximation and work with four-significant-digit accuracy until two successive iterates agree within 0.001 in each...
-
Why are intermolecular forces generally much weaker than bonding forces?
-
A widely used undergraduate experiment is the recrystallization of acetanilide from water. Acetanilide (see following structure) is moderately soluble in hot water, but much less soluble in cold...
-
Give the structure of each of the following compounds. (a) Chlorocyclopropane (b) Methylene iodide
-
Acetone (Table 8.2) has a significant dipole moment (2.7 D). Using structures, show the stabilizing interactions to be expected between acetone solvent molecules and (a) A dissolved potassium ion;...
-
The market price of a security is $50. Its expected rate of return is 10%. The risk-free rate is 5%, and the market risk premium is 8%. What will the market price of the security be if its beta...
-
At the end of 2025, Aramis Company has accounts receivable of $800,000 and an allowance for doubtful accounts of $40,000. On January 16, 2026, Aramis Company determined that its receivable from...
-
Your father is 50 years old and will retire in 10 years. He expects to live for 25 years after he retires, until he is 85. He wants a fixed retirement income that has the same purchasing power at the...

Study smarter with the SolutionInn App


