What products would you expect from the following reactions? CH2 (a) + CHCI3 CH (b) Zn(Cu)
Question:
What products would you expect from the following reactions?
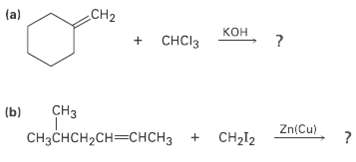
Transcribed Image Text:
CH2 (a) кон + CHCI3 CHз (b) Zn(Cu) CH2I2 снзснсH2сH—снсHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (19 reviews)
Strategy Reaction of a double bond with chloroform under basic conditions gives a product wi...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from the following reactions? (a) (b) (c) (d) (e) OMe OMe
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
What products would you expect from the following reactions? (a) (b) OCH3 A, 2 CuLi OCH3 Br + Ph-Culi-
-
What type of isomers are exhibited by [Fe(en) 3 ]Cl 2 (en = ethane-1,2-diamine)? no isomers are possible. cis and trans isomers fac and mer isomers optical isomers
-
Random samples of tires being replaced by a car dealer showed the tire life (miles) below, based on whether or not the owner had checked once a month for recommended tire inflation. Use = .05 in the...
-
sin(2). Simplify the answer in terms of cos(2). Use the angle addition formulas (Equations 2.10.1 and 2.10.2) to expand the following and use the product and sum rules to compute the derivatives of...
-
Know the reasons for using value management in projects.
-
Sadowski Video Center accumulates the following cost and market data at December 31. Compute the lower-of-cost-or-market valuation for Sadowskiinventory. Inventory Categories Cameras Camcorders DVDs...
-
Write a Java conditional statement that implements the following table, where grade is an integer and school is a String. Both variables have been declared and grade has been initialized to a value...
-
Maplebrook Spas provides the following data for the year just ended December 31st. Requirement Prepare the operating activities section of Maplebrook Spas' statement of cash flows for the year ended...
-
The following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structures of both, and explain the result.
-
What product would you obtain from catalytic hydrogenation of the following alkenes? CH2H (a) (b) CH "CH
-
A conference organizer decided to have eight separate groups of panelsA, B, C, D, E, F, G, and Hto make decisions on eight parallel tracks for a professional meeting. To facilitate communication...
-
Gitano Products operates a job - order costing system and applies overhead cost to jobs on the basis of direct materials used in production ( not on the basis of raw materials purchased ) . Its...
-
Martinez Corporation issues $630,000 of 9% bonds, due in 11 years, with interest payable semiannually. At the time of issue, the market rate for such bonds is 10 %. Click here to view factor tables....
-
Mickley Company's plantwide predetermined overhead rate is $18.00 per direct labor-hour and its direct labor wage rate is $11.00 per hour. The following information pertains to Job A-500: Direct...
-
Minor, Incorporated, had revenue of $ 5 7 2 , 0 0 0 and expenses ( other than income taxes ) of $ 2 8 2 , 0 0 0 for the current year. The company is subject to a 3 5 percent income tax rate. In...
-
Assume that payroll records of GG Shoes provided the following salary information for the weekly pay period ended December 1 3 , 2 0 2 4 : A B C D E F G H EMPLOYEE HOURS WORKED 4 0 HOURS WEEKLY...
-
What type of plate boundary is the San Andreas Fault?
-
What are some of the various ways to implement an awareness program?
-
A substance has a triple point at -24.5 C and 225 mmHg. What is most likely to happen to a solid sample of the substance as it is warmed from -35 C to 0 C at a pressure of 220 mmHg? (a) The solid...
-
Label each of the following molecules as a hydrogen-bond acceptor, donor, or both. Tndicate the hydrogen that is donated or the atom that serves as the hydrogen-bond acceptor. (a) (b) (c) H3c -...
-
Classify each of the following substance according to their solvent properties (as in Table 8.2.) (a) 2,2,2-triftuoroethanol ( - 26) (b) 2,2,4-trimethylpentane ( : 2)
-
The experiment is repeated, except that I mL of l-nonanol is added instead of methanol. In which layer is the alcohol dissolved? Explain.
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...
-
John Fleming, chief administrator for Valley View Hospital, is concerned about the costs for tests in the hospital s lab. Charges for lab tests are consistently higher at Valley View than at other...
-
A company is looking at new equipment with an installed cost of $436,859. This cost will be depreciated straight-line to zero over the project's 6-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


