How would you prepare the following alkyl halides from the correspondingalcohols? CH (a) CI (b) Br CH
Question:
How would you prepare the following alkyl halides from the correspondingalcohols?
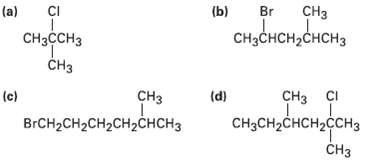
Transcribed Image Text:
CHз (a) CI (b) Br CнзссHз CHзснсH2CнсHз CHз (c) CI сHз снз (d) BrCH2CH2CH2CHгснCHз CHзCH2снсH2cсH3 CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Remember that halogen acids are used for converting tertiary alcohols to al...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
How would you prepare the following substances from Cyclopentanol? More than one step may be required. (a) Cyclopentanone (b) Cyclopentane (c) 1-Methylcyclopentanol (d) Trans-2-Methylcyclopentanol I...
-
How would you prepare the following compound using an acetoacetic estersynthesis?
-
Tiffee Company identifies the following items for possible inclusion in the physical inventory. Indicate whether each item should be included or excluded from the inventory taking. (a) 900 units of...
-
Consider the example of the new product at Curtis Distributors, which was analyzed using Holt's method in Figure 7.12. Repeat the forecasts using one smaller value of a than the value of 0.2 used in...
-
The cantilever beam AB shown in the figure is an S6 x 12.5 steel I-beam with E = 30 x 106 psi. The simple beam DE is a wood beam 4 in. x 12 in. (normal dimension) in cross section with E = 1.5 x 106...
-
How can an organization justify implementing a CRM system ?
-
1. Explain user-centered design principles in a brief memo to Annie. 2. Suggest at least four user interface design guidelines that could be used for the new system. 3. Suggest several types of...
-
Barbara Jackson and Fred Harrin decide to organize the Wildhorse partnership. Jackson invests $15,300 cash and Harrin contributes $10,200 cash and equipment with a cost of $7,140 and accumulated...
-
The Pharma Biotech Corporation spent several years working on developing a DHA product that can be used to provide a fatty-acid supplement to a variety of food products. DHA stands for...
-
What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures ofall. H (a) . (b) CH3D2H
-
How strong a base would you expect a Grignard reagent to be? Look at Table 8.1, and then predict whether the following reactions will occur as written.?(The p K a of NH 3 is 35.) (a) CH 3 MgBr + H ?...
-
In the 1990s, there was widespread adoption of just-in-time inventory techniques by U.S. businesses, which are techniques that companies use to keep inventories from running out or building up beyond...
-
Consider a bond with annual payments of $100, a principal payment of $1,000 in 10 years, and a cost of $1000. The Duration of the bond is 6.759 and the Convexity of the bond is 52.792, assuming a...
-
Boyd Crowder signed loan papers agreeing to borrow $40,000 at 4 percent compounded monthly. The first monthly loan payment of $625.81 is due one month from today. How many loan payments must Boyd...
-
You check your credit card balance, and notice that the interest rate is quoted as 20.6% APR. You also know that interest is compounded monthly. What is the Effective Annual Rate on your credit card?
-
A project that will provde annual cash flows of $2,800 for nine years costs $9,200 today. a. At a required return of 11 percent, what is the NPV of the project? b. At a required return of 27 percent,...
-
A mining company purchased $5 million in new construction equipment for a new project to begin in the current year. It is expected that the project will yield revenues of $10 million in the first...
-
How can you evaluate the effectiveness of training?
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
Write a molecular equation for the gas-evolution reaction that occurs when you mix aqueous nitric acid and aqueous sodium carbonate.
-
The cyclohexane chair just drawn has the headrest to the left and the footrest to the right. Draw a cyclohexane chair with its axial and equatorial bonds, having the headrest to the right and the...
-
Draw 1, 2, 3, 4, 5, 6-hexamethylcyclohexane with all the methyl groups (a) in axial positions. (b) in equatorial positions. If your cyclohexane rings look awkward or slanted when using the analytical...
-
Draw a Newman projection, similar to Figure 3-25, down the bond in the equatorial conformation of methylcyclohexane. Show that the equatorial methyl group is also anti to C5. (Using your models will...
-
A couple obtained a $20,000 mortgage loan at an interest rate of 10.5% compounded monthly. (Original principal equals to PV of all payments discounted at the interest rate on the loan contract) (1)...
-
What strategies and tactics are employed to manage strategic risks and uncertainties, including geopolitical instability, supply chain disruptions, and emerging competitive threats, while preserving...
-
How do strategic planners integrate ethical considerations and sustainability imperatives into strategic planning processes, balancing short-term financial objectives with long-term societal and...

Study smarter with the SolutionInn App


