What products would you expect from the followingreactions? ta) CH3CH2CH2C=CH + 2 Cl2 () (b) 1 HBr
Question:
What products would you expect from the followingreactions?
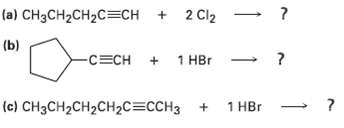
Transcribed Image Text:
ta) CH3CH2CH2C=CH + 2 Cl2 (ь) (b) 1 HBr -СЕCH + 1 нBr -C=CH c) CH3CH2CH2CH2C3СCHЗ + 1 HBr - ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Markovnikov addition is observed with alkynes as ...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from the reaction of ethylmagnesium bromide (CH3 CH2MgBr) with each of the following reagents? (a) H2O (b) D2O (c) (d) (e) (f) (g) then H O "H Ph" PhPh, then NH4CI, HO...
-
What products would you expect from the following reactions? (a) (b) (c) (d) (e) OMe OMe
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
Which subject does Corporate social responsibility belong to?
-
A firm is trying to decide whether to enter a highly uncertain market now or to wait to decide two years from now, when the size of the market will be less uncertain. If it enters now it must invest...
-
A 60o strain rosette, or delta rosette, consists of three electrical-resistance strain gages arranged as shown in the figure. Gage A measures the normal strain (a in the direction of the axis. Gages...
-
Briefly describe the three phases of the World Wide Webs development.
-
Roedel Electronics produces tablet computer accessories, including integrated keyboard tablet stands that connect a keyboard to a tablet device and holds the device at a preferred angle for easy...
-
Transportation of natural gas is commonly done via pipelinesacross long distances. A company uses a 0.6-m diameter pipe totransport natural gas. Then pumping stations are located atdifferent points...
-
Exhibit 1.18 represents common-size income statements and balance sheets for seven firms that operate at various stages in the value chain for the pharmaceutical industry. These common-size...
-
There are seven isomeric alkynes with the formula C6H10. Draw and name them.
-
What product would you obtain by hydration of the following alkynes? (a) CH3CH2CH2C=CCH2CH2CH3 CH (b) CCH2CECH-CH2CH3
-
In Exercises find an equation of the hyperbola. Vertices: (2, +3) Point on graph: (0,5)
-
A passenger transportation company estimates that next year it will sell $300,000 from the sale of 1,000 tickets. Each truck can carry 20 passengers and on each trip the truck costs $2,800 for...
-
Walker Bat Company is a private company that manufactures a line of high-end baseball bats. They have been in business 25 years and have a reputation in the industry for high-quality products and...
-
A company's inventory balance was $37,900 at the beginning of the year and $35,000 at the end. The inventory turnover ratio for the year was 5.2 and the gross profit ratio 35%. What were net sales...
-
The monthly fixed costs of a mini motel are $32000. The price per night for the coming year is set at $115. The costs that arise from rentals per day as the base-unit are $15 for maid service, $7 for...
-
Wildhorse Company has a balance in its Accounts Receivable control account of $11,200 on January 1, 2022. The subsidiary ledger contains three accounts: Bixler Company, balance $4,100; Cuddyer...
-
What is the purpose of the Concepts part of each chapter in the Principles volume of this book? What is the purpose of the Quantitative Tools part of each Principles chapter?
-
Design a circuit which negative the content of any register and store it in the same register.
-
Complete and balance each gas-evolution equation. a. HBr(aq) + NIS(s) b. NH4l (aq) + NaOH(aq) c. HBr(aq) + NaS(aq) d. HCIO4(aq) + LiCO3(aq)
-
The artificial sweetener aspartame (sidebar, p. 1208) was withheld from the market for several years because, on storage for extended periods of time in aqueous solution, it forms a diketopiperazine...
-
Identify each of the compounds A-D in the reaction scheme shown in Fig. P26.67, p. 1331. Explain your answers. H,N NH NaNO /HCI CH(CH3)2 HCL H20 C -heat (CHs)-CH-CH-=O + D
-
When peptides containing the Asn-Gly sequence, such as H in the equation given in Fig. P26.69, are stored in aqueous solution at neutral or slightly basic solution, ammonia is liberated and a...
-
Pretend you need to conduct a title search. You have the information needed from your online search, but you need to go to the clerk's office to finish your search. What information/records will/can...
-
Watch the Tedx Talk (Melvin Russell: I love being a police officer, but we need reform) by Lt. Colonel Melvin Russel from the Baltimore Police. In his talk, he discusses how law enforcement is in...
-
Solve for X. Assume X is a 2 x 2 matrix. Do not use decimal numbers in your answer. If there are fractions, leave them unevaluated. X = # 6 X+ 7 = 7 8 X. [24]*+41-41 -5

Study smarter with the SolutionInn App


