What reagent would you use to accomplish each of the following reactions? , 2. (a) CHCH-CH2co
Question:
What reagent would you use to accomplish each of the following reactions?
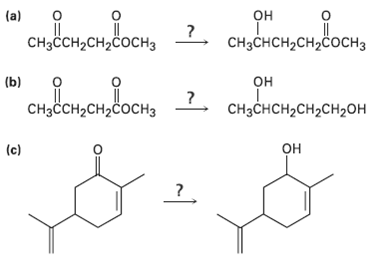
Transcribed Image Text:
обенодо, 2. ореородвонть (a) CHзснCH-CH2coснз CHзссH2CH2cосHз сононвоси, 2. он (b) CHзснсH2сH2сH2он CHзҫсH2сH2cосHз он (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
1 NaBH4 2 H30 OH CH3CHCHCHCOCH 3 NaBH4 reduces aldehydes and k...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What reagents would you use to accomplish a substitution with retention of configuration; for example? OH SH (R)-2-Butanol (R)-2-Butanethiol
-
Identify the reagent you would use to accomplish each of the following transformations: (a) Cyclobutanol bromocyclobutane (b) tert-Butanol tert-butyl chloride (c) Ethyl chloride ethanol
-
Each of the following reactions has been reported in the chemical literature and proceeds cleanly in good yield. Identify the principal organic product in each case. (a) (b) (c) (d) (e) (f) (g) (h)...
-
Alistair bought a house on 1 April 2000 for 125,000 and occupied the entire house as his principal private residence until 1 November 2008. As from that date, he rented out two rooms (comprising...
-
Why are short-term objectives needed when long-term objectives are already available?
-
Prove the second De Morgan Law.
-
Kent County Grain and Milling Company decided to stimulate the sale of its flour by including a coupon, redeemable for fifty cents ( \(50 q\) ), in every 25 - and 50 -pound sack of flour produced...
-
Selected transactions from the journal of Eberle Inc. during its first month of operations are presented here. Instructions (a) Post the transactions to T-accounts. (b) Prepare a trial balance at...
-
Determine the equivalent uniform annual cost of a process which will involve an initial outlay of $70,000 followed by costs of $8,000 in year 1, $9,000 in year 2, and amounts increasing by $1,000 per...
-
Create a class named Customer that will determine the monthly repayment amount due by a customer for a product bought on credit. The class has five (5) fields: customer name, contact number, product...
-
Predict the products of the following reactions: CH (a) 1. CHCH2- 2. NaOH, H2O2 "CH (b) 1. HglOAc)2. 0 2. NABH4 (c) CCH2CH2CH2 CH2CH2CH2CH3 C=C 1. Os04 2. NaHSO3, H20 -
-
What carbonyl compounds give the following alcohols on reduction with LiAlH4? Show allpossibilities. (a) CH- (b) CHCH3 (d) (CH)2HCH20 (c)
-
The Cobb-Douglas production function for a product is P = 5L 0.8 K 0.2 , where P is the quantity produced, L is the size of the labor force, and K is the amount of total equipment. Each unit of labor...
-
What are invasion of privacy issues usually deals with the unpermitted use of a person's name or likeness for another's benefit or advantage?
-
adiscuss the dynamic interplay between mitochondrial dynamics, bioenergetics, and cellular metabolism, and how disturbances in these processes contribute to the pathogenesis of metabolic disorders,...
-
What are the molecular mechanisms underlying the regulation of circadian rhythms at the transcriptional, translational, and post-translational levels, and how do disruptions in these processes impact...
-
Today, you sold 300 shares of Royal Bank of Canada (RY) stock. Your total return on these shares is 50.48%. You purchased the shares one year ago at a price of $80.72 per share. You received a total...
-
Imagine a market with a demand function of QD = 12 - P and a supply function of QSP 2. If the price is $4, find the consumer surplus (CS), producer surplus (PS), dead weight loss (DWL), and the...
-
Huang Automotive ended its fiscal year on July 31, 2025. The companys adjusted trial balance as of the end of its fiscal year is as follows. Instructions a. Prepare the closing entries using page...
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
Construct a concept map showing the ideas contained in a phase diagram.
-
What reaction will take place if ethyl alcohol is added to a solution of HCC:- Na+ in liquid ammonia?
-
Acid HA has pKa = 20; acid HB has pKa = 10. (a) Which is the stronger acid? (b) Will an acid-base reaction with an equilibrium lying to the right take place if Na+A- s added to HB? Explain your...
-
Which of the following are potential Lewis acids and which are potential Lewis bases? (a) (b) (c) (C6H5)3P: (d) (e) (CH3)3B (f) H: - CH.CH-N-CH CH3 CH3 H3C-C CH Br:
-
What choice is a multiple of 5? a) 93 b) 63 c) 65 d) 84
-
Determine how many tablets will be needed to give the dosage. Prepare a dosage of 6.4 mg using tablets with a strength of 1.6 mg.
-
(a). Company Inc., private consumer regulator firm, claims that the standard deviation of the rating of Formula One races in the town main car racing circuit was at least 15.34. (b). An outside...

Study smarter with the SolutionInn App


