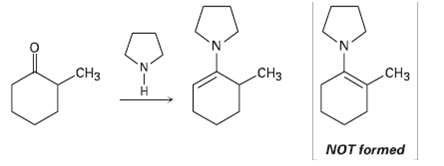
When 2-methylcyclohexanone is converted into an enamine, only one product is formed despite the fact that the
Question:
When 2-methylcyclohexanone is converted into an enamine, only one product is formed despite the fact that the starting ketone is unsymmetrical. Build molecular models of the two possible products, and explain the fact that the sole product is the one with the double bond away from the methyl- substitutedcarbon.
Transcribed Image Text:
CH3 CH3 CH3 NOT formed
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
CH CH3 Crowding between the methyl group and the py...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Despite the fact that market risk capital requirements have been imposed on FIs since the 1990s, huge losses in value were recorded from losses incurred in FIs trading portfolios. Why did this...
-
When treated with sodium borohydride, d-glucose is converted into an alditol. (a) Draw the structure of the alditol. (b) Which l-aldohexose gives the same alditol when treated with sodium borohydride?
-
Despite the fact that union membership is relatively low, many people believe the fear of unionization ensures that employers treat employees well. Do you agree or disagree with this view? Why?
-
When developing cost functions, which of the following statements is FALSE? A. Personal observations of costs and activities provide the best evidence of a plausible relationship between a cost and...
-
Is the extent of creative destruction likely to differ across industries? Can the risk of creative destruction be incorporated into a five-force analysis of an industry?
-
For orbital that are symmetric but not spherical, the contour representations (as in Figures 6.22 and 6.23) suggest where nodal planes exist (that is, where the electron density is zero). For...
-
What are the four deliverables of the IT strategic planning process?
-
Merit Bay Communications operates a customer call center that handles billing inquiries for several large insurance fi rms. Since the center is located on the outskirts of town, where there are no...
-
Overhead Application, Fixed and Variable Overhead Variances Zepol Company is planning to produce 600,000 power drills for the coming year. The company uses directl $1,777,500. The total fixed...
-
As the senior accountant at Active Quality Sportswear (AQS), which manufactures sport attire for men, women, youth, and children, you are often asked to prepare various financial analysis necessary...
-
The third and fourth steps in the synthesis of Hagemans ester from ethyl acetoacetate and formaldehyde are an intra molecular aldol Cyclization to yield a substituted Cyclohexanone, and a...
-
The Stork en amine reaction and the intra molecular aldol reaction can be carried out in sequence to allow the synthesis of Cyclohexanone. For example, reaction of the pyrrolidine enamine of...
-
Accountants for Carlson, Inc. have assembled the following data for the year ended December 31, 2016: Prepare Carlsons statement of cash flows using the indirect method. Include an accompanying...
-
Chi Hou's Noodles makes noodles by hand and supplies local Chinese restaurants. The company has the following standards for direct labour: Staff should be able to make 10 kilograms per hour and the...
-
The following four events affecting property, plant and equipment, and intangible assets, occurred during the current year for a firm: ( i . ) Purchased equipment for $ 5 0 , 0 0 0 . ( ii . )...
-
Given Information Initial Investment = $ 1 , 0 0 0 , 0 0 0 WACC = 1 0 % Revenue = $ 8 5 0 , 0 0 0 COGS = $ 5 4 0 , 0 0 0 Operating Expenses = $ 5 0 , 0 0 0 Depreciation Expense = $ 1 2 5 , 0 0 0 Tax...
-
On January 1 , 2 0 2 0 , Bank X makes a loan of $ 1 0 , 0 0 0 on a 1 year note at an interest rate of 1 0 % to a client. How much interest revenue is recognizable in total at the end of September 3 0...
-
At a local discount store 5 toasters ovens and 8 slow cookers cost $350 while 9 toaster ovens and 5 slow cookers cost $395. How much does one toaster oven cost? How much is one slow cooker? Cost of a...
-
The rotational inertia of any flat object about an axis perpendicular to the object is equal to the sum of the rotational inertias about two mutually perpendicular axes in the plane of the object, if...
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
Use covalent Lewis structures to explain why each element (or family of elements) occurs as diatomic molecules. a. Hydrogen b. The halogens c. Oxygen d. Nitrogen
-
In the hydroboration of 1-methylcyclopentene shown in Solved Problem 8-3, the reagents are achiral, and the products are chiral. The product is a racemic mixture of trans-2-methylcyclopentanol, but...
-
Predict the major products of the following reactions. Include stereochemistry where applicable. (a) 1-methylcycloheptene + BH3 THF, then H2O, OH- (b) Trans-4,4dimethylpent-2ene + BH THF, then H2O2,...
-
(a) When (Z)-3-methylhex-3-ene undergoes hydroboration-oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the...
-
A large hospital required nurses to work 80 hours over two-week (14 day) pay periods. In addition, nurses were also considered to be "on-call" for an additional 20 hours during the pay period. While...
-
Describe the history of the HIPAA, explaining why it is essential. Consider the following questions in your response: Why is understanding the history of the HIPAA law essential in decision making...
-
"I'm not sure we should lay out $265,000 for that automated welding machine," said Jim Alder, president of the Superior Equipment Company. "That's a lot of money, and it would cost us $75,000 for...

Study smarter with the SolutionInn App


