Which of the following alkenes would you expect to be good Diels-Alder dienophile?s? (b) (a) H2C=CHCI %3H2H2C
Question:
Which of the following alkenes would you expect to be good Diels-Alder dienophile?s?
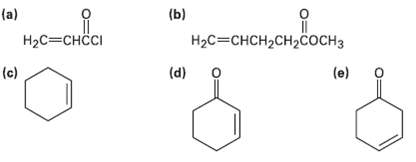
Transcribed Image Text:
(b) (a) H2C=CHCI Нас%3снсH2сH2Cоснз (d) (c) (e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Strategy Good dienophiles have an electronwithdrawing group conjugated with a double bond Good di...View the full answer

Answered By

Rohit anand
i have done btech from nit,hamirpur..i teach physics. i am teaching physics from last 2 year ..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Which of the following processes would you expect to be under control, and which would you expect not to be under control? Explain briefly why or why not. (a) Daily sales at each checkout line in a...
-
Which of the following nuclides would you expect to be radioactive: tungsten-184, polonium-206? Justify your choices. Ni, Cu, Ag, 47 108 Ag.
-
What is the square root of 3 to the square root of 2 power times the square root of 3 to the negative square root of 2 power?
-
How might a persistent global credit crisis affect the scale and scope of modern firms?
-
Calculate [OH-] and pH for each of the following strong base solutions: (a) 0.182MKOH, (b) 3.165 g of KOH in 500.0 mL of solution, (c) 10.0 mL of 0.0105 MCa(OH)2 diluted to 500.0 mL, (d) A solution...
-
Assume your audit plan requires observing at least one deviation at a specified critical rate of deviation, and the expected rate of occurrence is zero. Population size is 7,750, the critical rate of...
-
Strong Tool Company has been considering purchasing a new lathe to replace a fully depreciated lathe that will last 5 more years. The new lathe is expected to have a 5-year life and depreciation...
-
Use the following information from separate companies a through d Net Income (Loss) Interest Expense Income Taxes a. $ 182,000 $ 65,520 $ 45,500 b. 176,600 58,278 63,576 C. 154,700 d. 185,100 40,222...
-
1. Prepare a payroll register for Mary's Luxury Travel for the week ended March 22, 20--. (In the Taxable Earnings/Unemployment Compensation column, enter the same amounts as in the Social Security...
-
Predict the product of the following Diels-Alderreaction: - C=C
-
Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the s-trans dienes, which can readily rotate tos-cis? (a) (c) (b)
-
Find a normal vector n and an equation for the planes in Figures 8(A)(C). N (A) -3 (B) (C)
-
hat ways do modern operating systems contribute to energy efficiency and power management? Discuss techniques like Dynamic Voltage and Frequency Scaling (DVFS) and Advanced Configuration and Power...
-
How do operating systems ensure fault tolerance and recovery in case of system failures? Discuss the role of checkpoints, journaling, and redundancy in this context.
-
A substantial portion of inventory owned by Prentiss Sporting Goods was recently destroyed when the roof collapsed during a rainstorm. Prentiss also lost some of its accounting records. Prentiss must...
-
(Candy Company] Milton, Ava, and Hiro own a candy company. The business is failing and Milton is declared bankrupt by the bankruptcy court. Ava wants to keep the business running because she is sure...
-
What distinguishes a real-time operating system from a general-purpose operating system? Discuss the use cases for RTOS and the challenges in meeting real-time requirements.
-
You have to specify the power output of a motor for a ski tow rope that will carry 20 skiers at a time. The grade of the ski slope is \(32^{\circ}\) above horizontal, and the average coefficient of...
-
You deposit $10,000 in a savings account that earns 7.5% simple interest per year. What is the minimum number of years you must wait to double your balance? Suppose instead that you deposit the...
-
What are colligative properties?
-
Draw just the bonding -MO's for the cycloheptatrienyl cation. Draw the energy diagram to show the relative energies of all the MO's, and show which orbitals the electrons would occupy in the ground...
-
The proton NMR chemical shifts of the hydrogens in pyridine are shown. These are typical aromatic chemical shifts, except that the ortho protons (on the carbons bonded to nitrogen) are deshielded to...
-
Does the MO energy diagram of cyclooctatetraene (Figure 16-8) appear to be a particularly stable or unstable configuration? Explain. In Figure 16.8 nonbonding line cyclobutadiene cyclooctatetraene...
-
Discuss and describe transactional leadership in term of management. ?
-
How would each leadership style ( transactional leadership and transformational leadership) approach communication with various stakeholders?
-
Explained the different type of leader -Great man -trait theory -skill theory -behavior theory -Contingency theory (situational) -Transformation transactional leadership -transactional leadership...

Study smarter with the SolutionInn App


