Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the
Question:
Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the s-trans dienes, which can readily rotate tos-cis?
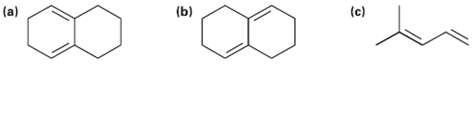
Transcribed Image Text:
(a) (c) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a This diene has an scis conformation and should underg...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following conjugated dienes would not react with a dienophile in a Diels-Alder reaction? a. b. c. d. e. f. CH CH7 CH2 CH2
-
Which of the following industries have low optimal debt levels according to the trade-off theory? Which have high optimal levels of debt? a. Tobacco firms b. Accounting firms c. Mature restaurant...
-
Which of the following reagents can oxidize H2O to O2(g) under standard-state conditions? H+(aq),Cl-(aq), Cl2(g), Cu2+(aq), Pb2+(aq), MnO4- (aq) (in acid).
-
The Cano Company is decentralized, and divisions are considered investment centers. Cano has one division that manufactures oak dining room chairs with upholstered seat cushions. The Chair Division...
-
Some firms seem to last forever. (For an extreme example, go to www.hbc.com.) In some industries, however, even the most effective firms may expect short lifetimes (e.g., lawn crews, Thai...
-
Use mesh analysis to find ix in Fig. 13.80, Where is = 4 cos(600t) A and vs = 110 cos(600t + 30º) 12 F 150 800 mH 600 mH
-
Product quality is of paramount importance in chemical product design. Explore the following quality improvement methodologies on the Internet: (a) Fishbone diagram (b) Pareto analysis (c) Process...
-
Refer to Matthias Medical's financial statements, presented in Exercise 12-8. Required Calculate the following market measure ratios for 2013. a. Earnings per share (average of 418,000 shares...
-
Discuss the thermodynamic principles underlying non-equilibrium processes, focusing on entropy production, irreversibility, and the role of transport phenomena in systems far from equilibrium, with...
-
Thunder Attractions, an amusement park, is considering a capital investment in a new exhibit. The exhibit would cost 136,000 and have an estimated useful life of 5 years. It will be sold for 60,000...
-
Which of the following alkenes would you expect to be good Diels-Alder dienophile?s? (b) (a) H2C=CHCI %3H2H2C (d) (c) (e)
-
Predict the product of the following Diels?Alder reaction: ?
-
Use the fact database and patron collection created in the text for use with MongoDB. Write a query to retrieve only the first name, last name, and type of faculty patrons that have checked out at...
-
Evaluate the integral TT/4 10 1 - sin0 cos0 do
-
Billy Bob is documenting the way data will be transformed from an existing system in order to become more compatible with the new system. What type of requirement is he documenting?
-
Take this file as input: state_population.csv ( I cant upload the file so I took a picture of the numbers) Use the population field as the datapoint to select your sample. Set the margin of error...
-
At a certain temperature, a 25.5-L container holds four gases in equilibrium. Their masses are 3.5 g.SO3, 4.6 g SO2, 14.3 g N2, and 0.98 g N2O. What is the value of the equilibrium constant at this...
-
2.2 A small closed cylinder has dimensions 50 mm external diameter by 160 mm long with 0.25 mm wall thickness. (a) What is the allowable internal pressure if the safe tensile stress for the material...
-
Find the inverse Laplace transform by using the partialfraction expansion method. \(\frac{4 s+5}{s^{2}\left(s^{2}+4 s+5 ight)}\)
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
Explain why the lower vapor pressure for a solution containing a nonvolatile solute results in a higher boiling point and lower melting point compared to the pure solvent.
-
Make a model of cyclooctatetraene in the tub conformation. Draw this conformation, and estimate the angle between the p orbitals of adjacent pi bonds.
-
Classify the following compounds as aromatic, antiaromatic, or nonaromatic. (a) (b) (c) (d)
-
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic. heptalene azulene pentalene
-
What choice is a multiple of 5? a) 93 b) 63 c) 65 d) 84
-
Determine how many tablets will be needed to give the dosage. Prepare a dosage of 6.4 mg using tablets with a strength of 1.6 mg.
-
(a). Company Inc., private consumer regulator firm, claims that the standard deviation of the rating of Formula One races in the town main car racing circuit was at least 15.34. (b). An outside...

Study smarter with the SolutionInn App


