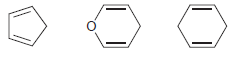
Which of the following compounds would you expect to be most acidic? Justify your choice.
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Deprotonatio...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds would you expect to show ultraviolet absorptions in the 200 to 400nmrange? (a) (b) (c) CN CH3 (f) (d) (e) " N. Indole Aspirin
-
Which of the following compounds would you expect to have a ? ? ?* UV absorption in the 200 to 400 nm range? (a) (b) (c) (CH3)2C=C=Do CH2 A ketene Pyridine
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
Use the trapezium rule, with interval-halving and extrapolation, to evaluate 0 log(cosh x) dx to 4dp
-
What is markup? How is it used to determine prices?
-
In a lawsuit against Angies List, Inc. for overtime pay, sales representatives were paid based on a 40-hour workweek but alleged that they routinely worked 50 to 60 hours a week without extra...
-
The Doak Company uses prenumbered vouchers. During a given test period, voucher numbers 1000 to 8000 were used. Required: a. Select the first five vouchers to be included in a random sample in each...
-
The Beranek Company, whose stock price is now $25, needs to raise $20 million in common stock. Underwriters have informed the firms management that they must price the new issue to the public at $22...
-
Suppose you have just joined an office of an accounting firm, MC Partners, that provides audit and various types of non-audit services to its clients. Within a few months of joining, you discover...
-
Dynamic Mattress decides to lease its new mattress stuffing machines rather than buy them. As a result, capital expenditure in the first quarter is reduced by $50 million, but the company must make...
-
What are some things Amazon does to manage inventories in their warehouses?
-
How can performance measures sometimes cause the wrong things to happen?
-
Who should you select for your systems-testing team? What skills should they have?
-
Due to the rapid growth of your technology company, the executive team has asked HR to develop an internal training program. The purpose of the program is to help employees recently promoted to...
-
1. A close corporation wants their shareholder agreement to indicate that shareholders can withdraw with six months' notice. What else should the transfer restrictions in the share agreement state?...
-
Roger, who works Darkstar Ltd is extremely proud of his invention and cannot wait to share it with his good friend Mark, who work for another firm. He takes the software and a prototype of the...
-
Students are expected to read the article referred through https://logisticallyspeaking.online/difference-between-3pl-and-4pl-their-advantages-and-disadvantages/. This article would also be discussed...
-
a. Assume Alan got into a car, knowing that it was not his, "hot wired" it, and then drove off in it. Is he guilty of violating the statute? Why? b. Assume Bill approached a car that he intended to...
-
Use the Midpoint Rule for triple integrals (Exercise 24) to estimate the value of the integral. Divide B into eight subboxes of equal size. SII, cos(xyz) dV, where B = {(x, y, z) | 0 < x < 1, 0 < y <...
-
Do public and private companies follow the same set of accounting rules? Explain.
-
(a) From what Grignard reagent can 3-methl-l pentanol be prepared by reaction with ethylene oxide, then aqueous acid? (b) Give the structure of another epoxide and another higher-order curpate that...
-
Explain why all attempts to isolate trimethyloxonium iodide lead instead to methl iodide and dimethl ether.
-
Explain why all attempts to isolate trimethyloxonium iodide lead instead to methl iodide and dimethl ether.
-
Find V 1 V 5 V source I 2 I 3 I 4 I 5 R 1 R 2 R 3 R 4 R total. V 2 = 6 0 . 0 V , V 3 = 9 0 . 0 V , V 4 = 5 0 . 0 V , I 1 = 1 . 5 A , I total = 4 . 0 A , R 5 = 3 0 ohms. Indicate where appropriate....
-
Two point charges, -2.4C and 5.616 C, are placed at x = 0 cm and x = 9.7 cm, respectively. Consider the x- axis directed to the right. 1.0p 8a At what point along the x axis is the electric field...
-
As you know, the value of. It is defined as the ratio of the circumference of a circle C divided by its diameter 2r. That is x-C/2. a) Let's assume that you measured a circumference of a circle to be...

Study smarter with the SolutionInn App


