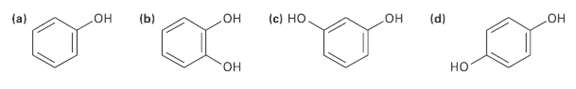
Which of the following molecules has a dipole moment? Indicate the expected direction ofeach. (b) (d) (c)
Question:
Which of the following molecules has a dipole moment? Indicate the expected direction ofeach.
Transcribed Image Text:
(b) (d) (c) Но Он Он Он (a) но Он но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a b CI ...View the full answer

Answered By

User L
I am a proficient writer with vast experience in medicine, nursing, biochemistry, microbiology, biology chemistry, accounting, economics, mathematics, statistics, actuarial science, psychology, sociology, philosophy, and social science. I deliver papers which are original, free from plagiarism, free from grammatical errors and high-quality in time
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following molecules has the largest a value: CH4, F2, C6H6, Ne?
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
The PF3 molecule has a dipole moment of 1.03 D, but BF3 has a dipole moment of zero. How can you explain the difference?
-
When a cosmetic manufacturer tests the market to determine how many women will buy eyeliner that has been tested for safety without subjecting animals to injury, is it involved in a descriptive...
-
Tyneka inherited 1,000 shares of Aqua, Inc. stock from Joe. Joe's basis was $35,000, and the fair market value on July 1, 2016 (the date of death) was $45,000. The shares were distributed to Tyneka...
-
Random samples of size n1 = 55 and n2 - 65 were drawn from populations 1 and 2, respectively. The samples yielded p1 = .7 and p2 = .6. Test H0: (p1 - p2) = 0 against Ha: (P1 - P2) > 0 using a = .05.
-
Evaluate revenue and expense balances in regard to materiality and audit risk.
-
(Application of Years-of-Service Method) Andrews Company has five employees participating in its defined-benefit pension plan. Expected years of future service for these employees at the beginning of...
-
The resistors in the circuit below have the following values: R = 7.70 2, R = 7.00 2, and R = 3.80 2. The two batteries each have a voltage of 5.00 V. ww R ww R (a) Find the current in through R3. A...
-
The following information was recorded for a highway two years ago. Since then, the traffic has increased by 15% per year and the % heavy vehicles has doubled each year. (4+2+2+1) Total vehicle...
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
(a) The H C1 bond length is 136 pm. What would the dipole moment of HC1 be if the molecule were 100% ionic, H+ C1-? (b) The actual; dipole moment HC1 is 1.08 D. What is the percent ionic character...
-
How useful is the adage, When in Rome, do as the Romans do for someone who wants to work in another country for a while?
-
Write a 5 -8 page essay in MLA style about "Drones" with references. including introduction, and thesis. To generate MLA citations, use CCC library, Purdue Owl; citethisforme.com: this is a citation...
-
Trying to put all my ideas together in paragraphs for part of an essay. 3 different solutions. 1ST SOLUTION The process of separating collected textiles into three broad categories: re-wearable,...
-
A company bought a new machine for its warehouse on January 1: Paid $10,000 in cash Financed the rest of the purchase price via a $30,000 5-year note Incurred a separate $2,000 setup charge Monthly...
-
If 52+4f(x)+3x (f(x)) = 0 and f(-4) = -1, find f'(-4). f'(-4)=-1
-
Evaluate the integral. 4x+5 So 2x2+5x+1 dx
-
A positively charged dust particle passes between the two oppositely charged plates of an electrostatic precipitator. There is an electric force directed toward the negative plate, but the drag force...
-
Imagine that your best friend knows you are taking a psychology course and wonders what psychology is all about. How would you define psychology for your friend? Write an essay on the discipline of...
-
A gas is continuously passed through an adiabatic turbine at the rate of 2 mol/s. Its initial temperature is 600 K, its initial pressure is 5 bar and its exiting pressure is 1 bar. Determine the...
-
Bromine is larger than chlorine, yet the two atoms have identical axial destabilization energies. Explain.
-
Draw the stereo isomers of these compounds: (a) 1, 3-Dimethyleyclohexane (b) 1, 2-Diethylcycloproane (c) 1-Chloro-3-methylcyclopentane
-
Draw both chair conformations of trans-1, 3-dimethyl cyclohexane indicate whether each methyl group is axial or equatorial.
-
Bailey Delivery Company, Incorporated, was organized in 2021 in Wisconsin. The following transactions occurred during the year: a. Received cash from investors in exchange for 17,000 shares of stock...
-
Write a program (C++) that prompts the user to input an integer and then outputs both the individual digits of the number and the sum of the digits. Example: Input: 3456 Output: 3 4 5 6 Sum of digits...
-
The soil profile at a site consists of 1.5 m of sand underlain by a 6 m thick clay layer. The water table coincides with the top of the sand layer. The saturated unit weight (Ysat) of the sand and...

Study smarter with the SolutionInn App


