Use the electro negatively table (Figure) to predict which bond in each of the following sets is
Question:
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound.
(a) H3C ? C1 OR C1 ? C1
(b) H3C ? H OR H ? C1
(c) HO ? CH3 OR (CH3)3Si ? CH3
(d) H3C ? Li OR Li ? OH
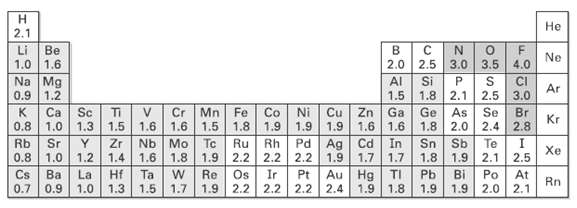
Transcribed Image Text:
Не 2.1 Li Be 1.0 1.6 B. 2.0 Ne 2.5 3.0 2.5 3.0 3.5 | 4.0 Na Mg 0.9 1.2 Ca 0.8 CI Ar Al 1.5 1.8 2.1 Si Ge As Ni Cr Mn Zn Ga Se Cu Br Kr Sc Ti 1.5 1.3 1.6 1.6 Zr Nb Mo 1.2 1.4 La Hf 1.5 1.7 1.0 1.3 Fe Co 1.8 1.9 Ru Rh Tc 1.6 1.6 In 1.7 Hg| TI 1.9 1.8 1.8 2.0 2.4 2.8 1.0 Rb 1.5 1.9 1.9 Pd Ag 1.6 1.8 | 1.9 2.2 | 2.2 2.2 Sr 0.8 Cd Sb Sn 1.8 Xe Te 1.0 Cs 1.7 2.5 1.9 2.1 1.9 Ba 0.7 0.9 Bi Po 1.9 2.0 2.1 1.9 Pb At Rn Re 1.9 Pt Au Ta Os Ir 2.2 | 2.2 2.2 2.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
a H3C CI 0 b More p...View the full answer

Answered By

Vinay Singh
I am master of science in physics and master of educationand have teaching experience of 18years.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Without using Fig. 13.3, predict which bond in each of the following groups is the most polar. a. COF, SiOF, GeOF b. POCl, SOCl c. SOF, SOCl, SOBr d. TiOCl, SiOCl, GeOCl e. COH, SiOH, SnOH f. AlOBr,...
-
Predict which substance in each of the following pairs would have the greater intermolecular forces. a. CO2 or OCS b. SeO2 or SO2 c. CH3CH2CH2NH2 or H2NCH2CH2NH2 d. CH3CH3 or H2CO e. CH3OH or H2CO
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
A teacher has just given an algebra exam. What are some of the statistics she could compute?
-
On December 28, 2016, Kramer sells 150 shares of Lavender, Inc. stock for $77,000. On January 10, 2017, he purchases 100 shares of the same stock for $82,000. a. Assuming that Kramer's adjusted basis...
-
Japanese researchers have developed a compression/depression method of testing electronic circuits based on Huffman coding (IEICE EEI Transactions on Information & Systems, Jan. 2005). The new method...
-
Indicate the specific applicability of the audit objectives to revenue and expense balances.
-
(Ethical IssuesCompensation Plan) The executive officers of Rouse Corporation have a performance-based compensation plan. The performance criteria of this plan are linked to growth in earnings per...
-
Why is specialization among workers necessary, especially in today's knowledge economy? But what are the risks of specialization and how can we mitigate them? Considering how a modern hospital is...
-
In Problem reverse the order of integration for each integral. Evaluate the integral with the order reversed. Do not attempt to evaluate the integral in the original form. 0 1 y2 4ye dx dy
-
Identify the most electronegative element in each of the following molecules: (a) CH2FC1 (b) FCH2CH2CH2Br (c) HOCH2CH2NH2 (d) CH3OCH2Li
-
Which of the following molecules has a dipole moment? Indicate the expected direction ofeach. (b) (d) (c) (a)
-
Rotations on spin states are given by an expression identical to Equation 6.32, with the spin angular momentum replacing the orbital angular momentum: In this problem we will consider rotations of a...
-
Explain the role of an HR audit as it relates to strategy formation. Describe one benefit of an HR audit
-
3. McClain, Edwards, Shiver, and Smith (MESS) LLC is considering the purchase of new automated cleaning equipment. The industrial engineer for the company, David "the Dirtman" R., has been asked to...
-
Write a 5 -8 paragraph essay in MLA styles about "Drones" with references. In order to generate MLA citations, use CCC library, Purdue Owl; citethisforme.com: this is a citation generator for MLA and...
-
Find dy/dx using implicit differentiation. You must show all your work for any credit. 2x+3y-7 In(x + y) ) Evaluate the derivative. You must show all you work for any credit. Your final answer must...
-
Kristen the first child, took the path of four year college with a full-time job upon graduation and started saving immediately. She faithfully put $100 a week into an investment account for the next...
-
Raindrops acquire an electric charge as they fall. Suppose a 2.0-mm-diameter drop has a charge of +12 pC; these are both very common values. In a thunderstorm, the electric field under a cloud can...
-
It is possible to investigate the thermo chemical properties of hydrocarbons with molecular modeling methods. (a) Use electronic structure software to predict cHo values for the alkanes methane...
-
Use the Estimation tool in Aspen Plus to estimate the physical properties of methyl vinyl ketone (MKK) after entering structure using the Molecular Structure tool.
-
Draw a Newman projection of the highest-energy conformation of 2,3-dimethylbutane about the C2-C3 bond.
-
Draw the two chair conformations for ethyl cyclohexane which is more stable.
-
Which of these compounds will have more of the conformation with the substituent on the cyclohexane ring axial present at equilibrium? CH,CH3 CI CH3 Ph CH,CH, b) or or or
-
Answer the following questions with respect to the following Strategy pattern diagram as illustrated below 3. The relationship between StudentToSchool class and Strategy and actors with strategy?...
-
a) Design a simple relational database that captures the information required to maintain records of orders. Take into account that your design should allow a customer to order as many items as...
-
Write a procedure named AVG in assembly language which calculates and returns the average of three memory operands. Also show how this procedure will be called from main. Pass arguments (variable...

Study smarter with the SolutionInn App


