Which of these compounds would have a faster rate of SN2 reaction? a) b) c) d) e)
Question:
Which of these compounds would have a faster rate of SN2 reaction?
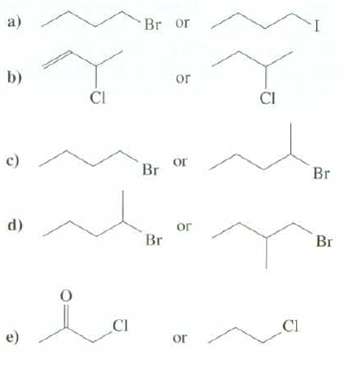
Transcribed Image Text:
a) b) c) d) e) CI CI Br or or Br or Br or or CI CI Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
a The right compound has a faster rate because iodide ion is a better leaving group than bromid...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which compound has a faster rate of SN2 reaction: a) HC CI CI c) PhCHCH3 or H CI Cl or CHCHCH3 b) CI CH3 CH3 or U
-
Which of these compounds would have faster rate of SN1 reaction? a) b) c) d) e) CI ta Ph Ph -CI or Br or Br or or -Cl or + CH3 Ph -Cl Br Br ta -CI
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
A long cylindrical wood log (k = 0.17 W/mK and = 1.28 10 -7 m 2 /s) is 10 cm in diameter and is initially at a uniform temperature of 15C. It is exposed to hot gases at 550C in a fireplace with a...
-
Steelworx Ltd accumulates costs for its single product using weighted average process costing. Direct material is added at the beginning of the production process, and conversion occurs uniformly...
-
The following information is from the books of OZP Farms, Inc. regarding its employee stock options. The firm granted options on January 2, 2015, that permit employees to acquire 100,000 shares of $...
-
What is the relationship between a Problem Space and an Opportunity Space?
-
The Hudson Corporation makes an investment of $24,000 that provides the following cash flow: YearCash Flow 1 .........$ 13,000 2 ......... 13,000 3 ......... 4,000 a. What is the net present value at...
-
Suppose that we have two machines A and B. The following table shows the execution times for programs that make up a benchmark suite for machine A, machine B, and a reference machine X. Program Exec...
-
Locate all the instantaneous centres for the crossed four bar mechanism as shown in Fig. 6.28. The dimensions of various links are : CD = 65 mm; CA = 60 mm ; DB = 80 mm ; and AB = 55 mm. Find the...
-
(a) Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
Arrange these compounds in order of increasing SN2 reaction rate: CI Br CI Br
-
Find the equation of the parabola described. Find the two points that define the latus rectum, and graph the equation. Focus at (-4, 0); vertex at (0, 0)
-
A manager of Gregorio Radio Station is facing a problem between competing strategies (both would benefit the company). Specifically, the business has Php 500,000 to invest the money between 2...
-
Given the set of images above, choose two images and explain how it is being related to managerial economics.
-
The line tangent to the graph of f(x) = sin x at (0,0) is y=x. What does this imply? Choose the correct answer below. A. The line y = x is the best straight line approximation to the graph of f for...
-
5. A study was conducted on the amount of time drivers wait for a stoplight to change at a particular intersection. The amount of time spent by 300 drivers was recorded and the resulting data were...
-
Choose... Scenario Analysis Payback Period Forecasting Risk/Estimating Risk Security Market Line (SML) Capital Gains Yield Risk Equation Project Net Working Capital Breakeven Internal Rate of Return...
-
What is an illegal market? Under what circumstances do illegal markets arise?
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
Draw a box-and-whisker plot for each data set. 4 3 Hits in a Round of Hacky Sack 12 3 3 5 18 1 19 6 3
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither. (a) NABH4 H20 CH;CH- CH3CH2CH2OH (b) OH 1. BH3 2. NaOH, H202
-
Give a JUPAC name for each of the following alkyl halides (yellow green =Cl): (b) (a)
-
How could your cultural models influence your ability to learn your students' funds of knowledge and utilize those funds? How could you or will you address this?
-
Bella deposits $300 per month into an account paying 6% nominal interest compounded monthly for 3 years, to be used to purchase a car. Which equation should she use to compute the price of the most...
-
Assume that management wants wage concession from its counterpart labor union. Explain why this often leads to disputes. Suppose management gives up some of its future negotiating power by subjecting...

Study smarter with the SolutionInn App


