Write a mechanism for the conversion of -ketoglutarate to succinyl CoA in step 4 of the citric
Question:
Write a mechanism for the conversion of -ketoglutarate to succinyl CoA in step 4 of the citric acid cycle(Figure).
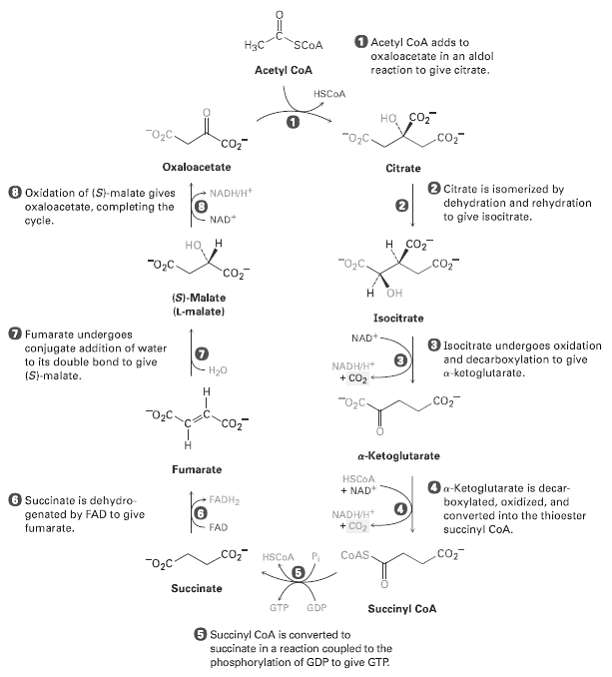
O Acetyl CoA adds to oxaloacetate in an aldol H3C SCOA Acetyl CoA reaction to give citrate. HSCOA HO co, .co co2 Oxaloacetate Citrate O Citrate is isomerized by dehydration and rehydration to give isocitrate. NADHUH O Oxidation of (St-malate gives oxaloacetate, comploting the O NAD cycie. н сот но "O,C. co, Co H OH (S)-Malate (L-malate) Isocitrate O Fumarate undergoes conjugate addition of water to its double bond to give NAD+ O lsocitrate undorgoes oxidation and decarboxylation to give a-kotoglutarato. NADHH H20 (S-malate. + Co2 H. co, co, a-Ketoglutarate Fumarate HSCOA Oa-Ketoglutarate is decar- boxylated, oxidized, and converted into the thioester + NAD O Succinate is dehydro genated by FAD to give fumarate. FADH2 NADHH + Co, FAD succinyl CoA. CO HSCOA .co, COAS. Succinate GTP GDP Succinyl CoA 6 Succinyl CoA is converted to succinate in a reaction coupled to the phosphorylation of GDP to give GTP.
Step by Step Answer:
The steps in the conversion of aketoglutarate to succinyl CoA are simila...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Organic Chemistry questions
-
Write a mechanism for the following reaction. OH H SO. H2O
-
Write a mechanism for the following reaction. cat. H.SO OMe
-
Write a mechanism for the following radical halogenation reaction. Br Br2 hi
-
International Markets Project You have been hired as a globalization specialist by Kwik-E-Mart. Kwik-E-Mart is a large retailer that has outgrown its domestic market and seeks to expand its...
-
The purpose of this practice exercise is to illustrate how a change in an input cell impacts variables on other financial statements. Using the Excel spreadsheet model in the file folder entitled...
-
The tensile strength of nonferrous pipes is of importance. Samples of size 5 are selected from the process output, and their tensile strength values are found. After 30 such samples, the process mean...
-
Consider the hemodialysis device using the hollow-fiber module in Figure 24.1, operating \(12 \mathrm{hr} / \mathrm{wk}\) (3 days, \(4 \mathrm{hr} /\) day) at a dialysis center. As shown in Example...
-
Modern Company acquires the net assets of Frontier Company for $1,300,000 on January 1, 2011. A business valuation consultant arrives at the price and deems it to be a good value. Part A. The...
-
What are the consequences of a poor person-job fit? What are the consequences of a poor person-organization fit? What are the benefits of hiring the right people for the right job and what are the...
-
The sequence number field in the TCP header is 32 bits long, which is big enough to cover over 4 billion bytes of data. Even if this many bytes were never transferred over a single connection, why...
-
Another step in the pentose phosphate pathway for degrading sugars is the conversion of ribose 5-phosphate to glyceraldehydes 3-phosphate. What kind of organic process is occurring? Propose a...
-
In step 2 of the citric acid cycle (Figure), cis-aconitate reacts with water to give (2R, 3S)-isocitrate. Does ?OH add from the Re face of the double bond or from the Si face? What about ?H? Does the...
-
We have an accounting standard that requires the disclosure of information about operating segments. What is an operating segment?
-
-5 a) Calculate 2 = (-23+2i) and write the answer Cartesian form. on b) solve the equation 2+(4+2i)z+ (12-4) 0 +H
-
What is the output of the given code block? = 10 a b = 20 C = 30 if a % b > 1: print(a * b) elif b % a == 10: print (b a) print(c % 11) ww
-
19. Construct the flowchart and algorithm of the following problem: A fishing fleet fishes in 10 different regions each consisting of 8 different areas. The data about the fish caught in kilograms...
-
Given the graph of y = f(x) below, answer all of the following questions. -5 1 y 5 Activa
-
What are the definitions for these words? administrative law, cause of action, civil law, compensatory damages, constitutional law, contract, demurrer, depose, due diligence, felony, laws,...
-
1. Does vertical integration matter if consumers still have a choice of suppliers and if generators are still competing with each other? 2. Wholesale and retail energy markets are expected to deliver...
-
Outline some of the major problems confronting an international advertiser.
-
Redo Problem 14.2 using Aspen Plus. Problem 14.2 Compute the flame temperature of an oxyacetylene torch using pure acetylene and 50 percent more pure oxygen than is needed to convert all the...
-
Write the structure of the major organic product formed in the reaction of hydrogen chloride with each of the following: (a) 2-Methyl-1-butene (b) cis-2-Butene (c) CH3CH
-
All the following reactions have been reported in the chemical literature. Give the structure of the principal organic product in each case. (a) (b) (c) (d) (e) (f) (g) (h) (i) no peri CH,CH CH CHCH...
-
A single epoxide was isolated in 79-84% yield in the following reaction. Was this epoxide A or B? Explain your reasoning. CH;COOH B.
-
In the Heckscher-Ohlin model, as trade occurs in a capital-abundant country, increased imports will force domestic import- competing firms to decrease prices and production. Labor and capital will...
-
Why has Gatorade been so successful in the sports drink market?
-
Mohali Foods has estimated that fixed costs per month are Rs . 1 1 4 , 8 4 0 and variable costs per unit of sales is 4 2 % . For the month the business anticipates sales of Rs . 3 0 0 , 0 0 0 . What...

Study smarter with the SolutionInn App


