How would you carry out the following reactions to introduce deuterium into organicmolecules? (a) CHH2C3CCH2C Hs 2H5
Question:
How would you carry out the following reactions to introduce deuterium into organicmolecules?
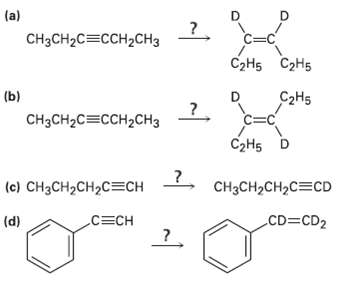
Transcribed Image Text:
(a) CHзсH2C3CCH2CНз СэHs С2H5 (b) C2H5 CHзсH2C3CCH2CH3 C=C СэHs D (c) CH3CH2CH2C=CH CH3CH2CH2C=CD CD CD2 C=CH (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
a b c d CH3CHCCCHCH3 CH3CHCCCHCH3 CH3CH...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
YOU have just graduated from Deakin as an accounting and finance graduate. During your time at Deakin, you were organized and diligent with your studies and thus graduated with flying colors. This...
-
A manufacturer of nylon carpets produces rolls of carpeting at four factories and ships them to distributors in five locations The following table shows the capacities at the factories and the...
-
In a recent basketball game, a player who makes 65% of his free throws made eight consecutive free throws. Assuming that free-throw shots are independent, determine whether this feat was unusual.
-
1.15 Euro Virtual's EPS Sensitivity to Exchange Rates (B). Assume a major weather crisis hits Switzerland, reducing its agricultural and food industries and sub- sequently leading to a macroeconomic...
-
MBTA Corporation issued bonds and received cash in full for the issue price. The bonds were dated and issued on January 1, 2011. The stated interest rate was payable at the end of each year. The...
-
Do you think the value of a company is impacted by its capital structure and What are the auditor's responsibilities for inventory maintained in public warehouses or with other outside custodians?...
-
Suppose the total benefit derived from a continuous decision, Q, is B(Q) = 20Q 2Q2 and the corresponding total cost is C(Q) = 4 + 2Q2, so that MB(Q) = 20 4Q and MC(Q) = 4Q. a. What is total benefit...
-
How would you synthesize the following compounds from acetylene and any alkyl halides with four or fewer carbons? More than one step may herequired. (a) CH3CH2CH2C=CH (b) H2C%3CCH CH CH CHH2H3D (d)...
-
How would you prepare Cyclodecyne starting from acetylene and any alkyl halide needed?
-
For the same work team you used in Field Exercise 6-35, examine copies of any relevant written documentation (e.g., written procedures, forms, reports, system documentation). Are any of these forms...
-
31. Find the center, the vertices, and the foci. A) (x+3)+(y-5) = 1 25 25 36 B) 3(x+2)2+4(y-1) = 192
-
Calculate the monthly mortgage payment. Round to the nearest cent. Answer $120,000 is financed for 20 years at 4.25 % APR
-
What are the primary mechanisms by which catalysts can deactivate in a chemical reactor, and how can these be mitigated in the design of a long-term industrial process ?
-
18. [0/2 Points] DETAILS PREVIOUS ANSWERS TANAPCALCBR10 3.6.060. MY NOTES ASK YOUR TEAC Blowing Soap Bubbles Carlos is blowing air into a spherical soap bubble at the rate of 9 cm/sec. How fast is...
-
12 -19x+90 Find the limit, if it exists: (Do not use L'Hospital's rule) lim 29 22-26 +153 Note: Give the exact answer but not the decimal approximation(for example, write 4/5 istead of 0.8)....
-
The full-wave rectifier circuit shown in Figure 2.5 (a) in the text is to deliver \(0.2 \mathrm{~A}\) and \(12 \mathrm{~V}\) (peak values) to a load. The ripple voltage is to be limited to \(0.25...
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
What is the molarity of Cl in each solution? a. 0.200 M NaCl b. 0.150 M SrCl 2 c. 0.100 M AlCl 3
-
The following compounds are listed in increasing order of acidity. In each case, the most acidic proton is shown in red. (a) Show the structure of the conjugate base of each acid, including any...
-
Amides such as acetamide are much weaker bases than amines, such as ethylamine (CH3CH2NH2). (a) Use resonance forms to show why the nonbonding electrons on the nitrogen atom of the amide are very...
-
Methyllithium (CH3Li) is often used as a base in organic reactions. (a) Predict the products of the following acid-base reaction. CH3CH2 - OH + CH3 - Li (b) What is the conjugate acid of CH3Li?...
-
give answer and explain step by step solution . Check my work Bandar Industries manufactures sporting equipment. One of the company's products is a football helmet that requires special plastic....
-
. Pepper Pty Ltd makes and sells two types of shoes, Comfort and Style. Product data is as follows: Comfort Style Unit selling price $40.00 $45.00 Variable costs per unit$32.00 $34.50 Sixty per...
-
. Edge Company produces two models of its product with the same machine. The machine has a capacity of 138 hours per month. The following Information is available Standard Deluxe Selling price pe...

Study smarter with the SolutionInn App


