Show both the substitution and elimination products that are formed in these reactions: a) C CI +
Question:
Show both the substitution and elimination products that are formed in these reactions:
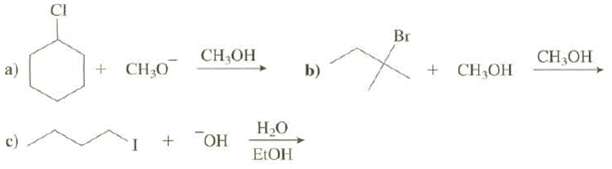
Transcribed Image Text:
a) C CI + CH₂0 CH₂OH + OH H₂O EtOH b) Br + CH₂OH CH3OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Elimination reactions compete with substitution reaction...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show both the substitution and elimination products that would be formed in these reactions: to , -CI a) Br CH,OH CH,O b) CI E:OH + CH;CH,OH
-
Give the substitution and elimination products you would expect from the following reactions. (a) 3-bromo-3-ethylpentane heated in methanol (b) 1-iodo-1-methylcyclopentane heated in ethanol (c)...
-
What substitution and elimination products (if any)might be obtained when each of the following alkyl halides is treated with sodium methoxide in methanol? (a) methyl iodide (b)...
-
Describe, in human terms, why delay and jitter are bad in real time (interactive) voice and video communications. Would these same problems apply to recorded voice and video stored and played back at...
-
The Launceston Wool Company designs and manufactures woollen coats, which are in high demand the cold Tasmanian winters. The company uses a process costing system to cost products because 1: produces...
-
Describe the differences in interest payments and bond price between a 5 percent coupon bond and a zero coupon bond.
-
\(\left(\frac{2}{5} \times\left(\frac{7}{8}-\frac{2}{3} ight) ight)^{2} \div\left(\frac{4}{9}+\frac{5}{6} ight)+\frac{7}{12}\) Perform the indicated operations. Reduce to lowest terms.
-
A manufacturing company has two service and two production departments. Building Maintenance and Factory Office are the service departments. The production departments are Assembly and Machining. The...
-
Barry and Pat are both approaching retirement. Full retirement age for each of them is age 66. Barry plans to file for Social Security benefits at 70 at which time Pat, who also will be 66 next year,...
-
A soft drink bottling company is interested in controlling its filling operation. Random samples of size 4 are selected and the fill weight is recorded. The table below shows the data for 24 samples....
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the rearranged carbocations that are expected from these carbocations: a) +CH CH3 b) CHCHCHCHCH c) CH3
-
The controller of MingWare Ceramics Inc. wishes to prepare a cost of goods sold budget for September. The controller assembled the following information for constructing the cost of goods sold...
-
Montoure Company uses a periodic inventory system. It entered into the following calendar-year purchases and sales transactions. Date January 1 February 10 March 13 March 15 August 21 September 5...
-
Let f(x) = x-1 and g(x) 9(x) = Find X (a) (fog)(x) (b) (gof)(-1) (c) (fof)(x) (d) (gog)(5)
-
Convert from IEEE 754 to Decimal (Double Precision) 00110100001100110...00
-
Water bottle in a hot car. In the American Southwest, the temperature in a closed car parked in sunlight during the summer can be high enough to burn flesh. Suppose a bottle of water at a...
-
Q8. The Greeks of a given call are Delta Gamma Vega = .7 = .45 = .0125 8.1 Employing this call and the put on the same underlying stock and with the same K and T as the call, trader JD creates the...
-
The op-amp bias currents for the circuit in Figure P14.38 are equal at \(I_{B 1}=\) \(I_{B 2}=1 \mu \mathrm{A}\). (a) Find the worst-case output voltages \(v_{O 1}\) and \(v_{O 2}\) for \(v_{I}=0\)....
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
Evaluate each using the values given. (-(-5-x)); use x = 6, and = = -2
-
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid...
-
Give IUPAC names for the following alkylhalides: (b) (a) CHCH2CH2CH21 H C (c) C,2I BrCH2CH2CH2CH2Br CH (e) Br (1) 1 -CH2CI CI CH (d) CH2H2CI CHH2H CHCH2CH2CH I
-
Draw structures corresponding to the following IUPAC names: (a) 2-Chloro-3, 3-dimethylhexane (b) 3, 3-Dichloro-2-methylhexane (c) 3-Bromo-3-ethylpentane (d) 1, 1-Dibromo-4-isopropylcyclohexane (e)...
-
models in healthcare that apllies to communication and socio - economic influences
-
ezto.mheducation.com/ext/map/index.html?_con=con&external_browser=0&launchUrl=https%253A%252F%252Fnewconnect.mhe xam 2 (Chapters 6 & 7) 10. Problem (2 parts) 10 2 Dints 00:57:28 Saved You loaned...
-
Research for a bond mutual fund offered here in the Philippines A. Name of theFund B. The composition of the Fund C. When is the Inception Date of the Fund D. Explain the current net asset value per...

Study smarter with the SolutionInn App


