Show the rearranged carbocations that are expected from these carbocations: a) +CH CH3 b) CHCHCHCHCH c) CH3
Question:
Show the rearranged carbocations that are expected from these carbocations:
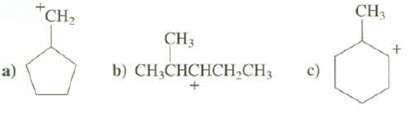
Transcribed Image Text:
a) +CH₂ CH3 b) CH₂CHCHCH₂CH₂ c) CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
A carbocation may rearrange to form a more stable ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show that a conditionally convergent series can be rearranged so to diverge?
-
Carbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secndary. Primary and methyl cations are too unstable to form in solution.
-
The acid-catalyzed dehydration of an alcohol to a rearranged alkene is known as a Wagner-Meerwein rearrangement. Propose a mechanism for the following Wagner-Meerwein rearrangement: OH ...wr + H30+...
-
1. What is Ladures target market and retail strategy in the United States? 2. Explain the reasons Ladure owns its stores in some countries and uses franchising with local licensees in others. 3....
-
Orbital Industries Ltd manufactures a variety of materials and equipment for the aerospace industry. A team of P & D engineers in the firm's Technology Park plant has developed a new material that...
-
A corporate bond with a 6.5 percent coupon has 15 years left to maturity. It has had a credit rating of BBB and a yield to maturity of 7.2 percent. The firm has recently gotten into some trouble and...
-
\(\frac{3}{8} \times\left(\frac{4}{9}-\frac{1}{8} ight)^{2}\) Perform the indicated operations. Reduce to lowest terms.
-
The Broughton Cap Company requires that prenumbered receiving reports be completed when purchased inventory items arrive in the receiving department. At the time of receipt, the receiving clerk...
-
Long Ltd., a private corporation adhering to ASPE, enters into a non-cancellable lease agreement on July 1, 2020, to lease equipment from Fong Ltd. The following data are relevant to the lease...
-
A car driver is travelling at speed v on a straight road. He comes over the top of a hill to find a fallen tree on the road ahead. He immediately brakes hard but travels a distance of 60 m at speed v...
-
Show both the substitution and elimination products that are formed in these reactions: a) C CI + CH0 CHOH + OH HO EtOH b) Br + CHOH CH3OH
-
Show the substitution products for these reactions: a) b) +CHCHOH Br CI Ja + HO EtOH HO EtOH
-
Suppose that the weather in Charlotte is modeled using the Markov chain in Section 10.1, Exercise 28. About how many days elapse in Charlotte between consecutive rainy days? Data From Section 10.1...
-
Performance Products Corporation makes two products, titanium Rims and Posts. Data regarding the two products follow: Direct Labor- Hours per unit Rims Posts 0.70 0.70 Annual Production 26,000 units...
-
Bond valuation-Semiannual interest Calculate the value of each of the bonds shown in the following table, all of which pay interest semiannually. (Click on the icon here in order to copy the contents...
-
(20%) A Fabry-Perot resonant cavity consists of a thin glass plate that has a refractive index of n = 1.50 and a thickness of = 100 m. Its surfaces are coated such that its peak transmittance is 100%...
-
For the financial year ended 31 March 2020, the draft final accounts of Langsam Berhad and its subsidiary, Schnell Berhad are as follows: Statement of Profit and Loss for the year ended 31 March 2020...
-
Determine the financial effect on the balance sheet and income statement for each of the following independent events using the Transaction Analysis Template. a . Paid $ 4 , 0 0 0 for six months...
-
(a) For the integrator circuit in Figure P14.48, let the input bias currents be \(I_{B 1}=I_{B 2}=0.1 \mu \mathrm{A}\). Assume that switch \(S\) opens at \(t=0\). Derive an expression for the output...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Solve each inequality and graph its solution. -3b + 5 + 7 < 14-b -9-8-7-654-3 -2 -1 0 1
-
Draw and name all monochloro products you would expect to obtain from radical chlorination of 2-methylpentane. Which, if any, are chiral?
-
Taking the relative reactivities of 1 o , 2 o , and 3 o hydrogen atoms into account, what product(s) would you expect to obtain from monochlorination of 2-methylbutane? What would the approximate...
-
Draw three resonance forms for the cyclohexadienylradical. Cyclohexadienyl radical
-
Kinesthetic, Read/Write and Aural are all learning modalities. True False
-
The correlation coefficient measures the: A.rate of return of individual stocks. B.direction of movement of the return of individual stocks. C.degree to which the returns of two stocks move together....
-
Which would you prefer, where your discount rate is = 8.5%? A.$12,000 now B.$1,800 per year for 10 years C.$8,000 now and $6,000 in two years D.$15,000 in 3 years please show work

Study smarter with the SolutionInn App


