Consider the following compounds and formulas. (The formulas are written in such a way as to give
Question:
Consider the following compounds and formulas. (The formulas are written in such a way as to give you an idea of the structure.)
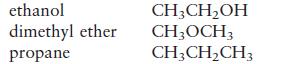
The boiling points of these compounds are (in no particular order) \(-42.1^{\circ} \mathrm{C},-23^{\circ} \mathrm{C}\), and \(78.5^{\circ} \mathrm{C}\). Match the boiling points to the correct compounds.
Transcribed Image Text:
ethanol dimethyl ether propane CH3CHOH CH3OCH3 CH3CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Matching Boiling Points to Compounds Heres the matching of boiling points to the correct compounds B...View the full answer

Answered By

Sufiyan Ahmed Tariq
I am a Chartered Accountant and an Associate Public & Finance Accountant. I also hold a bachelors of Commerce degree. I have over 8 years of experience in accounting, finance and auditing. Through out my career, I have worked with many leading multinational organisation.
I have helped a number of students in studies by teaching them key concepts of subjects like accounting, finance, corporate law and auditing. I help students understanding the complex situation by providing them daily life examples.
I can help you in the following subject / areas:
a) Accounting;
b) Finance;
c) Commerce;
d) Auditing; and
e) Corporate Law.
4.90+
7+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Are there dangers with incarcerating young people in detention facilities? What about the potential for negative long-term impact? Using three documented examples of misconduct/mistreatment of youth...
-
A corporation has two divisions (the Eastern Division and the Western Division) that operate semiautonomously, with each developing and marketing its own products. However, to coordinate their...
-
c++. error: array must be initialized with a brace enclosed initializer main.cpp X 10 11 ii 12 13 14 4567 15 16 17 18 19 287288285 20 21 23 24 25 26 27 28 25285922223288 30 31 33 34 36 37 40 41 42 43...
-
A tank contains 1000 L of brine with 15 kg of dissolved salt. Pure water enters the tank at a rate of 10 L/min. The solution is kept thoroughly mixed and drains from the tank at the same rate. How...
-
Unbilled Accounts Receivable Mike and Cary repair computers for small local businesses. Heavy thunderstorms during the last week of June resulted in a record number of service calls. Eager to review...
-
Draw four spanning trees of Graph \(B\) which includes edge \(u t\), but not \(u r\). Use Graphs \(A, B\), and \(C\). Graph A Graph B Graph C
-
Osage Inc. has actual sales for June and July and forecast sales for August, September, October, and November as follows: Actual: June . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Camden Corporation has provided the following contribution margin format income statement. Assume that the following information is within the relevant range. Sales ( 1 0 , 0 0 0 units ) $ 1 7 5 , 0...
-
Some of the physical properties of \(\mathrm{H}_{2} \mathrm{O}\) and \(\mathrm{D}_{2} \mathrm{O}\) are as follows: Account for the differences. (D is a symbol often used for \({ }^{2} \mathrm{H}\),...
-
For which molecule in each of the following pairs would you expect the stronger intermolecular forces? a. \(\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{NH}_{2}\) or \(\mathrm{H}_{2}...
-
Empirical research cited in the text indicates that firms with an operating cash flow to current liabilities ratio exceeding 40 percent portray low short-term liquidity risk. Similarly, firms with an...
-
1. a. How do different stakeholders view corporate social responsibility (CSR)? (Define corporate social responsibility, then answer through your definition) b. Give an example of competing...
-
If you were in charge of the social media campaign for the opening of new fast casual restaurant in your local area, what would be the key principles of social media engagement you would follow? What...
-
Post a description of the differences between change champions and opinion leaders. Be specific and provide examples. Then, describe the human resources that may be necessary to support your proposed...
-
what ways do individual differences in cognitive styles, such as analytical versus intuitive thinking, impact decision-making processes within complex organizational contexts, and how can these...
-
What is employee fraud? How can companies use control activities to mitigate the risk of employee fraud? INCLUDE ANY REFFERENCE USE
-
Suppose your company sells services for $325 cash this month. Your company also pays $100 in wages, which includes $15 that was payable at the end of the previous month and $85 for wages of this...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Hydrogen peroxide, H 2 O 2 , is a nontoxic bleaching agent being used as a replacement for chlorine in industry and home laundries. The bleaching process is an oxidation, and when hydrogen peroxide...
-
Hydroxylamine, HONH 2 , is used to remove hair from animal hides and as a photoresist stripper in the electronics industry. Describe the structure of the hydroxylamine molecule in terms of hybrid...
-
Estimate the bond angles marked with arcs and lowercase letters in peroxyacetylnitrate, an eye irritant in smog: HC a C N=0 b 0-0 Peroxyacetylnitrate
-
Calculate cost per unit. ( Round answer to 2 decimal places, e . g . 2 . 2 5 . ) Cost per unit $ eTextbook and Media Solution Attempts: 3 of 3 used ( b 2 ) Determine ( 1 ) the ending inventory and (...
-
Required: 1 . Calculate the following risk ratios for 2 0 2 5 . ( Use 3 6 5 days in a year. Round your intermediate calculation decimal place. ) Pls help asap
-
Exercise 3 - 1 A ( Algo ) Equation method LO 3 - 1 Adams Corporation produces products that it sells for $ 1 2 each. Variable costs per unit are $ 9 , and annual fixed costs are $ 6 1 , 2 0 0 . Adams...

Study smarter with the SolutionInn App


