Some of the physical properties of (mathrm{H}_{2} mathrm{O}) and (mathrm{D}_{2} mathrm{O}) are as follows: Account for the
Question:
Some of the physical properties of \(\mathrm{H}_{2} \mathrm{O}\) and \(\mathrm{D}_{2} \mathrm{O}\) are as follows:
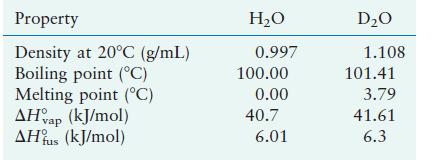
Account for the differences. (D is a symbol often used for \({ }^{2} \mathrm{H}\), the deuterium isotope of hydrogen.)
Transcribed Image Text:
Property Density at 20C (g/mL) Boiling point (C) Melting point (C) AHvap (kJ/mol) AHus (kJ/mol) HO 0.997 100.00 0.00 40.7 6.01 DO 1.108 101.41 3.79 41.61 6.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The differences in the physical properties between H2O water and D2O heavy water can be accounted fo...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The formula for shallow-water wave propagation speed, Eq. (10.9) or (10.10), is independent of the physical properties of the liquid, i.e., density, viscosity, or surface tension. Does this mean that...
-
When training a group of workers, often the challenge for the trainer is ensuring the key messages are getting through to everyone. In larger groups, its not always easy keeping everyone engaged, and...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
Consider the following molecule: Tin II Chloride(SnCl2) Are the bonds polar or non-polar? Is the molecule polar or non-polar?
-
A tank contains 1000 L of pure water. Brine that contains 0.05 kg of salt per liter of water enters the tank at a rate of 5 L/min. Brine that contains 0.04 kg of salt per liter of water enters the...
-
Royces Communication Sales Co. began operating in August. Royces does not offer discounts (all terms are net 30 days), and 8% PST and 5% GST are charged on all sales. Cost of inventory is calculated...
-
Label each of the following characteristics of a corporation as either an (A) advantage or a (D) disadvantage: a. Limited liability b. Taxation c. Regulations d. Transferability of ownership
-
A pet food company has a business objective of expanding its product line beyond its current kidney- and shrimp-based cat foods. The company developed two new products, one based on chicken liver and...
-
A small pilot study is run to compare a new drug for chronic pain to one that is currently available. Participants are randomly assigned to receive either the new drug or the currently available drug...
-
The unit cell for a pure xenon fluoride compound is shown in the following diagram. a. What is the formula of the compound? b. The unit cell of \(\mathrm{XeF}_{2}\) has a height of \(702...
-
Consider the following compounds and formulas. (The formulas are written in such a way as to give you an idea of the structure.) The boiling points of these compounds are (in no particular order)...
-
Write as a single faction. a. b. c. 1 1 V3 + 1 V3 1
-
In 2009, it was reported that a Mo-Mo quintuple bond (10, 2T, and 25 bonds) could be formed by reducing a quadruple-bonded dimolybdenum amidinate complex with KC8. Ar. Ar Ar- 4- Li[HC(N-2, 6-...
-
Describe Blue Apron's current marketing strategy (offline/online media and referral program) and its obstacles (rising CAC, decreasing retention, shifts in trends).
-
10) If the matrix [2 g] has only one eigenvalue, then a = - 86
-
Wimble Ltd had $400 million of debt outstanding at an interest rate of 9% and $600 million of equity (market value) outstanding. Wimble is subject to a 30% corporate tax rate. What is the amount of...
-
The stock of Alpha Tool sells for $81.20 per share. Its current dividend rate, D 0 , is $3 per share. Analysts and investors expect Alpha to increase its dividends at a 20 percent rate for each of...
-
The following matrix contains the costs ( in dollars) associated with assigning Jobs A, B, C, D, and E to Machines 1, 2, 3, 4, and 5. Assign jobs to machines to minimizecosts. 5-09292 137871 3-20367...
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
When iron surfaces are exposed to ammonia at high temperatures, nitridingthe incorporation of nitrogen into the iron latticeoccurs. The atomic radius of iron is 124 pm. (a) Is the alloy interstitial...
-
How would the height of a mercury barometer be different on the planet Mars? Explain your reasoning.
-
Air is a source of reactants for many chemical processes, such as the synthesis of ammonia. To determine how much air is needed for these reactions, it is useful to know the partial pressures of the...
-
For a one dimensional random walk the probability density function W(x) gives the distribution of possible outcomes after N random jumps each of length : W(x)=122e(x)2/22 . where is the average x...
-
Problem 11-04 (Replacement Analysis) Question 2 of 6 Check My Work Replacement Analysis Although the Chen Company's milling machine is old, it is still in relatively good working order and would last...
-
(3 points) Describe how you would compute the cross-validation mean squared error (us- ing the leave-one-out version of cross-validation) for the above model. In your description, make sure to...

Study smarter with the SolutionInn App


