Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the
Question:
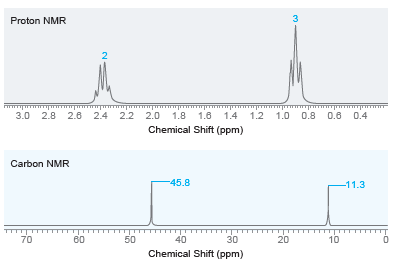
Transcribed Image Text:
3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4 Chemical Shift (ppm) Carbon NMR -45.8 -11.3 40 10 50 70 80 30 20 Chemical Shift (ppm) 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR and 13 C NMR spectra: Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber...
-
Perhaps more surprising to Mr. Pitkin was a proposal by the VP of Marketing to make a major investment in market share by increasing promotional expenditures by $2.5 million during 1998-2000. Sales...
-
Without calculating, determine whether the value of nPr is greater than the value of nCr for the values of n and r given in the table. Complete the table using yes (Y) or no (N). Is the value of nPr...
-
Mullins Distribution markets CDs of numerous performing artists. At the beginning of March, Mullins had in beginning inventory 2,500 CDs with a unit cost of $7. During March, Mullins made the...
-
What types of qualitative information should be disclosed about a company's leases?
-
Clark Kent, Inc., buys crypton for $0.80 a gallon. At the end of processing in Dept. 1, crypton splits off into products A, B, and C. Product A is sold at the split-off point with no further...
-
. An investor writes a December put option with a strike price of $47. The price of the option is $4. The investor makes a gain if the price of the stock is above $_ at the time of exercise. (This...
-
Your brother has just started a new job as the Controller of an IESBA restricted audit client. You do not serve on the audit engagement. What steps must you take to ensure your independence is not...
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Propose a mechanism for the following process: REN heat + N2 + CO2
-
The following information relates to two married couples: Compute the 2016 tax due or refund due for each couple. Assume that the itemized deductions have been reduced by the applicable floors....
-
Beth, an employer, wants to hire someone for a strenuous job that Beth, an employer, wants to hire someone for a strenuous job that requires a great deal of training, which will take place over the...
-
What are the determining factors for macroscopic observations? Explain
-
How should VinFast launch its eSUVs in Canada? - Market Trends - Competitive Analysis - Who's the target customer? - Product - Price - Promotion Place You need to write a marketing research report...
-
What are the underlying sociopolitical dynamics that drive social movements, and how do these movements impact societal norms and public policies ?
-
The city is issuing bonds to raise money for a building project. You obtain a $8200 bond that pays 9% simple interest annually that matures in 6 years. How much interest will you earn?
-
(a) When computing the standard deviation of a set of scores, when do we divide by N, and when do we divide by N 1? (b) What is the difference between and s?
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
(a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane. (b) Explain why free-radical halogenations usually gives mixtures of products. (c) How could...
-
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
otal compensation includes both direct and Blank______ compensation. Multiple choice question. intangible tangible external indirect
-
Transfer prices ____." Group of answer choices "are an accounting device to allocate the costs and revenues of intermediate products across divisions of a firm" "increase the 'profits' of the profit...
-
Costs not associated with replacing the services of a nonworking spouse include: Multiple choice question. the cost of a housekeeper or childcare provider the cost of a laundry service lost wages for...

Study smarter with the SolutionInn App


