Propose a mechanism for the following process: REN heat + N2 + CO2
Question:
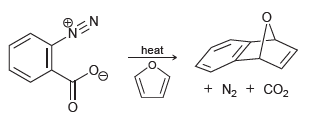
Transcribed Image Text:
REN heat + N2 + CO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
NEN Off R...View the full answer

Answered By

Balram Kumar Pandey
I have been teaching students for more than 5 years, my strongest subject is mathematics. In a span of 2 months I have completed mathematics of 10th class. Those students cleared the board exams.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for the following reaction. H2SO IM
-
Propose a mechanism for the following reaction. excess HBr Br tetrahydrofuran 1,4-dibromobutan
-
Propose a mechanism for the following ring-opening transesterification. Use the mechanism in Problem 21-13 as a model. H+ CH3OH HO OCH,
-
Let n=102 +r, where , re N, 0r9. A number a is chosen at random from the set {1, 2, 3, ..., n} and let pn denote the probability that (a 1) is divisible by 10. 70. If r = 0, then npn equals (a) 22...
-
1. The number of letter pairs that can be formed in any order from any two of the first 13 letters in the alphabet (A-M) is an example of a permutation. 2. The number of permutations of n elements...
-
Nixon Wholesale Corp. uses the LIFO cost flow method. In the current year, profit at Nixon is running unusually high. The corporate tax rate is also high this year, but it is scheduled to decline...
-
What determines whether a lease modification results in a separate lease or in a modification of an existing lease?
-
Two Indiana state senate candidates must decide which city to visit the day before the November election. The same four citiesIndianapolis, Evansville, Fort Wayne, and South Bendare available for...
-
1) Team Sports has 4.1 million shares of common stock outstanding, 2.1 million shares of preferred stock outstanding, and 21 thousand bonds. If the common shares are selling for $3.10 per share, the...
-
As a group you are the senior teaching staff of a school where each subject department is regarded as a cost centre. The direct costs of each cost centre are teachers salaries, books and worksheets...
-
Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the following 1 H NMR and 13 C NMR spectra: 3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4...
-
Phenacetin was widely used as an analgesic before it was removed from the market in 1983 on suspicion of being a carcinogen. It was widely replaced with acetaminophen (Tylenol), which is very similar...
-
In which of the following molecules would you expect the oxygen-to-oxygen bond to be the shortest? Explain. (a) H 2 O 2 , (b) O 2 , (c) O 3 .
-
Standard Deduction Amounts Filing Status 2020 Single 12,400 Married Filing Jointly or Surviving Spouse 24,800 Head of Household 18,650 Married Filing Separately 12,400 Child Tax Credit (per child)...
-
Maplewood Supply received a $5,350 invoice dated 4/15/20. The $5,350 included $350 freight. Terms were 2/10, 1/30, n/60. (If more than one discount, assume date of last discount.) If Maplewood pays...
-
Kellogg Company manufactures cereal and other convenience food under its many well-known brands such as Kellogg's, Keebler, and Cheez-It. The company, with over $13.5 billion in annual sales...
-
We have two unexpected problems at Calvert Crossings - both of which need to be addressed as soon as possible. First, we received a default notice on behalf of Foozer's Grille regarding a failure of...
-
How do various social institutions, such as the family, education, and the state, reinforce prevailing power structures within society ?
-
Use your statistical package to create a new variable that adds 50 points to the math quiz score. How have the measures of variability changed for this new variable as compared to the same measures...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
Rank the following carbocations in decreasing order of stability. Classify each as primary, secondary, or tertiary. (a) The isopentyl cation, (b) The 3-methyl-2-butyl cation, (c) The 2-methyl-2-butyl...
-
Each of the following proposed mechanisms for the free-radical chlorination of methane is wrong. Explain how the experimental evidence disproves each mechanism. (a) Cl2 + hv Cl2* (an activated form...
-
Rank the following radicals in decreasing order of stability. Classify each as primary, secondary, or tertiary. (a) The isopentyl radical, (b) The 3-methyl-2-butyl radical, (c) The 2-methyl-2-butyl...
-
Department F had 4,000 units in work in process that were 40% completed at the beginning of the period at a cost of $14,100. During the period, direct materials were added at a cost of $26,500, and...
-
During an IT audit, the auditor finds that loan customers are unable to log in to their online profiles to correct information in an application. Which privacy principle is most affected? Disclosure...
-
Select all that apply On 2/1, Sophie Company sold merchandise on account to Lexie Company in the amount of $1,500. Lexie Company paid the amount due on 2/28. The journal entry that Sophie Company...

Study smarter with the SolutionInn App


