1-Chioro-1, 2-diphenylethane can undergo E2 elimination to give either as- or trans-1, 2-diphenylethylene (stilbene). Draw Newman projections...
Question:
1-Chioro-1, 2-diphenylethane can undergo E2 elimination to give either as- or trans-1, 2-diphenylethylene (stilbene). Draw Newman projections of the reactive conformations leading to both possible products, and suggest a reason why the trans alkene is the majorproduct.
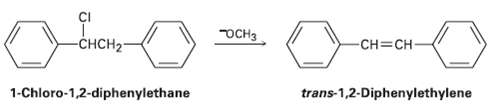
Transcribed Image Text:
осна -снCH2- -CH=CH- trans-1,2-Diphenylethylene 1-Chloro-1,2-diphenylethane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
H IA H Ph do Ph Ph Ph Ph OCH3 A H Both Newman project...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Suggest a reason why the proofreading step in protein synthesis takes place at the level of amino acid activation rather than that of codonanticodon recognition.
-
Draw Newman projections of the four stereo-isomers of 2-bromo-3-methylpentane in the conformation required for E2 elimination. Are the reactive conformations also the most stable conformations?...
-
Suggest a reason why many squatter settlements are built on floodplains or steep slopes.
-
In December 2014, Shire Computer's management establishes the 2015 predetermined overhead rate based on direct labor cost. The information used in setting this rate includes estimates that the...
-
Suppose you manage a mentoring program for your employer, and a well-liked manager tells you she receives so many requests to be a mentor that she isn't sure hot to handle them fairly. How could you...
-
1. The average distance in kilometers from a school in Chicago to the closest fast food restaurant is 0.60 with a standard deviation of 0.45. Express each of the following distances as a z score: a....
-
A company issues a \(10 \%\) coupon bond that matures in 5 years. However, this company is in trouble, and it is estimated that each year there is a probability of .1 that it will default that year....
-
At the end of Hotai Department Stores fiscal year on December 31, 2020, these accounts appeared in its adjusted trial balance. Freight-In ..........................................................NT$...
-
I. Byte stuff the following frame ESC Flag II. ESC ESC ESC Flag Bit unstuff the following received frame 01111110010001111101100111110010001111101111101001111101111110 III. Specify whether the...
-
It seems that some countries like China may be developing hybrid HRM systems. What are some of the advantages and disadvantages of the hybrid HRM system?
-
In addition to not undergoing substitution reactions, the alkyl bromide shown in Problem 11 .45 also fails to undergo an elimination reaction when treated with base. Explain.
-
Predict the major alkene product of the following E1reaction: CHBr eat CH-CH
-
Pizza sales and price, part 2. For the data in Exercise 31, the average Sales was 52,697 pounds (SD = 10,261 pounds), and the correlation between Price and Sales was = 0.547. If the Price in a...
-
1. You are given a circular wheel along with equipment with which the wheel can be fixed either with horizontal axis or with vertical axis. You also have access to any equipment that you have seen or...
-
What would be the voltage on a 4700uF capacitor 3.2 seconds after a 2602 resistor were connected across its terminals, assuming that the capacitor initially had a voltage of 12V on it?
-
A bicycle with 0.33 m radius wheels accelerates from rest to 9.67 m/s in a distance of 13.6 m with no slipping of the tires. Through what angle (in radians) do the wheels turn in this interval? rad...
-
An order is written as 25,000 units of heparin in 250 ml D5W to infuse at 1300 units/h, what is the correct rate of infusion (in ml/h)? How long will the current bag last, assuming the rate stays the...
-
Fawn Hollow LLC, is owned equally by three individuals, Mark Crenshaw, Thomas Miller, and Jerald Wolfowicz. The LLC was formed in 2014 for the purpose of building and operating a hotel property with...
-
Suppose that \(X\) is a continuous random variable that takes on values in \((0,1)\) and has characteristic function \(\psi(t)=[\exp (i t)-1] / i t\). Use Theorem 2.28 to find the density of \(X\)....
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this textbook. Instructions (a) Does East plats report any investments on its statement of...
-
Use the reactions and their equilibrium constants to predict the equilibrium constant for the reaction 2 A(s) 3 D(g). A(s) = 3 D(g) B(g) + C(g) B(g) + 2 C(g) K = 0.0334 K = 2.35
-
Propose a mechanism for the aldol condensation of cyclohexanone. Do you expect the equilibrium to favor the reactant or the product?
-
Give the expected products for the aldol condensations of (a) Propanal (b) Phenylacetaldehyde (c) Pentan-3-one
-
(a) Show each step in the mechanism of the acid-catalyzed interconversion of (R)- and (S)-3-methylpentan-2-one. (b) When cis-2,4-dimethylcyclohexanone is dissolved in aqueous ethanol containing a...
-
Zach Ltd. produced the following data for the year 2015 - o Interest Income $4,650,000 o Capital Gain realized $50,000 o Dividend Income from company's 15% ownership in XYZ Ltd. $65,000 o Corporate...
-
A project requires an increase in net working capital of $150,000 at time 0 that will be recovered at the end of its 10 year life. If the opportunity cost of capital is 8%, what is the effect of NWC...
-
A firm is 40% financed by debt with a yield-to-maturity of 8.5%. The equity has a beta of 1.3, the market risk premium is 8.4% and the risk-free rate is 3.8%. What is the firm's WACC if the tax rate...

Study smarter with the SolutionInn App


