In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2
Question:
In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, andwhy?
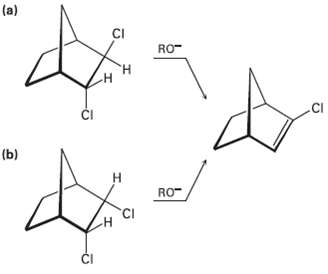
Transcribed Image Text:
(a) CI RO- н .CI ČI (b) Н RO- CI ČI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (19 reviews)
H N H H A B We concluded in Problem 1161 that E2 elimination i...View the full answer

Answered By

Ashish Jaiswal
I have completed B.Sc in mathematics and Master in Computer Science.
4.90+
20+ Reviews
39+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In light of your answer to Problem 14.43, propose a mechanism for the followingreaction: CH CH + CO2 Heat "CCH do,c, H a-Pyrone CHC-
-
In light of your answer to problem 11.42, what product might you expect from treatment of 4-hrorno-l-butanol withbase? Base BrCH2CH2CH2CH2
-
In light of your answer to Problem 4.44, draw the two chair conformations of 1, 1, 3-trimcthylcyclohexanc, and estimate the amount of strain energy in each. Which conformation is favored?
-
Adam Kleen Enterprise (AKE) was initially set up as a convenient shop selling laundry related items by Adam Bollan. Despite having good results in the national-level examination and was even offered...
-
How could a well-designed training program help make this idea meet ethical as well as legal standards?
-
1-Bromobicyclo [2.2.1] heptane is extremely unreactive in either SN2 or SN1 reactions. Provide explanations for this behavior.
-
Which of the following cash transfers misstates cash at December 31, 1999? Chooses the correct answer. Interbank Transfers Disbursement Recorded in books Receipt Paid by bank Recorded in books...
-
Donahue Company uses both special journals and a general journal as described in this chapter. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in...
-
A stone is thrown horizontally at 28.0 m/s from the edge of a cliff. The stone lands 46.0 m from the base of the cliff. Determine the time the ball was in the air.
-
Although anti periplanar geometry is preferred for E2 reactions, it isn?t absolutely necessary. The deuterated bromo compound shown here reacts with strong base to yield an un-deuterated alkene....
-
There are eight diastereomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane. Draw each in its more stable chair conformation. One isomer loses HCI in an E2 reaction nearly 1000 times more slowly than the...
-
Table shows that the percentage of children in families with income below the poverty line far exceeds the percentage of the elderly in such families. How might the allocation of government money...
-
A 83-gram sample of water that has an initial temperature of 8.0 C absorbs 6722 joules. If the specific heat of water is 4.184 J/(g C), what is the final temperature of the water? You must show all...
-
A block attached to a spring of the spring constant k = 500 N/m is pulled to stretch the spring by 0.40 m from the equilibrium point. Next moment, the block is released. When the block passes the...
-
When a horse pulls a 85 kg buggy down the road with a force of 38 N, the buggy hitch makes an angle of 35 degrees above the horizontal. Neglecting friction, what is the magnitude of the buggy's...
-
An ice skater with moment of inertia 70.0 [kgm2] is spinning at 41.0 rpm.( Note: 1 rpm = 1 revolution per minute.) If the skater pulls in her arms, her moment of inertia decreases to 50.0 [kgm2]....
-
The market risk premium for next period is 7.55% and the risk-free rate is 1.16%. Stock Z has a beta of 0.97 and an expected return of 10.93%. What is Stock Z's reward-to-risk ratio?
-
Marissa's spaceship approaches Joseph's at a speed of \(0.99 c\). As Marissa passes Joseph, they synchronize their clocks to both read \(t=0 \mathrm{~s}\). When Marissa's clock reads \(100...
-
In what ways does a well-designed enterprise search software vary from popular search engines (e.g., Bing, DuckDuckGo, and Google)?
-
Many equilibrium calculations involve finding the equilibrium concentrations of reactants and products given their initial concentrations and the equilibrium constant. Outline the general procedure...
-
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
-
1. Rank the following compounds in order of increasing acidity. 2. Indicate which compounds would be more than 99% deprotonated by a solution of sodium ethoxide in ethanol. () (b) () (d) () (f)...
-
Predict the products of the following aldol condensations. Show the products both before and after dehydration. (a) (b) (c) (d) (e) (f) CH3 TOH CH CH2-C-H CH TOH Ph-C-CH+ OH
-
1. Answer the following questions: Suppose you are given the following tabular dataset: Code Title Pages Study Level Price Availability 525745 Introduction to Java 256 Easy 57.20 Yes 525746 Advanced...
-
What is wrong with this code & why? How do I fix it? public class JavaApplication1 { private int num; public static void main(String[] args) { num = 14; num = num ++ % 3 + num; System.out.println(...
-
A classifier was built to distinguish between 3 types of news articles: S, H, and P. After training, it was run over 15 testing news articles and here are the results: Article # Actual type Predicted...

Study smarter with the SolutionInn App


