Amino acids, from which proteins are formed, exist as ?dipolar ions.? The structure of the dipolar ion
Question:
Amino acids, from which proteins are formed, exist as ?dipolar ions.? The structure of the dipolar ion of the amino acid alanine is
(a) Calculate the formal charges on all of the atoms, except hydrogen?s of alanine.
(b) What is the overall charge of alanine?
(c) Explain whether or not you expect the two oxygen?s to be different.
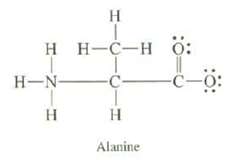
Transcribed Image Text:
H T HH-C-HÖ: H-N- H -C- I H Alanine -C-0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 86% (15 reviews)
a b The overall charge of ala...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Explain whether or not the following particles have an acceleration: (a) A particle moving in a straight line with constant speed and (b) A particle moving around a curve with constant speed.
-
In the previous exercise, explain whether or not you believe the sample is representative of the population of interest and why. In order to investigate how many hours a day students at their school...
-
The Illinois State University Alumni Association is having two Baseball Day events this year. The purpose of the events is to both foster relationships between ISU alums and to also raise money for...
-
The Real life highlights the risks to manufacturers of requiring suppliers to provide inventory on a just-in-time basis. In your own words, explain the advantages and disadvantages ix suppliers if...
-
The baggage limit for an airplane is set at 100 pounds per passenger. Thus, for an airplane with 200 passenger seats, there would be a limit of 20,000 pounds. The weight of the baggage of an...
-
What are the underlying basic concepts of a random sample?
-
Several years ago, Westmont Corporation developed a comprehensive budgeting system for profit planning and control purposes. While departmental supervisors have been happy with the system, the...
-
In order to feasibly implement the solution, analyze what would need to be considered. For instance, what resources (people, financial, etc.) would be involved? Or, what internal and/or external...
-
A plate 100 mm wide and 10 mm thick is to be welded with another plate by means of transverse welds at the ends. If the plates are subjected to a load of 70 kN, find the size of weld for static as...
-
Show a Lewis structure for NO 2 , both oxygens are bonded to the nitrogen.) Show a resonance structure also.
-
A covalent ion can also have polar bonds. Consider the ammonium caution. I low arc its bonds polarized? Do you think that the N of the ammonium caution is more or less ?electro negative? than the N...
-
In Molecular Systems Biology (Vol. 3, 2007), geneticists at the University of Naples (Italy) used reverse engineering to identify genes. They calculated P(G | D), where D is a gene expression data...
-
5.15 Water is added to the tank shown in Fig. P5.15 through a verti- cal pipe to maintain a constant (water) level. The tank is placed on a horizontal plane which has a frictionless surface....
-
A shaft carries four rotating masses A, B, C and D which are completely balanced. The masses B, C and D are 50 kg, 80 kg and 70 kg respectively. The masses C and D make angles of 90 and 195...
-
Estimate the yield stress of the iron crystal, when the iron crystal includes random dislocation network whose density is p=1.0 x 108cm/cm. The average length of the dislocation / is approximately...
-
2- For two-dimensional laminar jet flow, simplify the boundary layer equation and obtain relationship for p and q using the following stream function. 3- For flow with velocity profile: U(x)= 1.0+2.0...
-
Determine the maximum load for a simply supported beam of 5 0 ?mm diameter and 6 0 0 mm span, centrally loaded, as the load cyclically varies from W to 3 W . \ sigma en = 3 5 0 ?MPa, \ sigma yp = ...
-
Explain how emissions trading schemes are likely to affect financial reporting.
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
Graph the function f(x) = sin 50x in an appropriate viewing rectangle.
-
Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which iswhich? CH CH- CH CH2OH H3C CH3 H- CH Vitamin A (retinol) Vitamin C...
-
Fill in the multiple bonds in the following model of naphthalene, C10H8 (gray = C, ivory =H). How many resonance structures does naphthalenehave?
-
The following model is a representation of ibuprofen, a common over-the-counter pain reliever. Indicate the positions of the multiple bonds, and draw a skeletal structure (gray = C, red = O, ivory...
-
Pronghorn Corp reported net sales $675,000, cost of goods sold $438,750, operating expenses $154,000, and net income $69,525. Calculate the profit margin and gross profit rate. (Round Profit margin...
-
Victory Company uses weighted average process costing. The company has two production processes. Conversion cost is added evenly throughout each process. Direct ma added at the beginning of the first...
-
Here are selected 2022 transactions of Oriole Company. Jan. 1 June 30 Dec. 31 Retired a piece of machinery that was purchased on January 1, 2012. The machine cost $64,000 and had a useful life of 10...

Study smarter with the SolutionInn App


