Assign as many of the resonances as you can to specific carbon atoms in the 13C NMR
Question:
Assign as many of the resonances as you can to specific carbon atoms in the 13C NMR spectrum of ethylbenzoate.
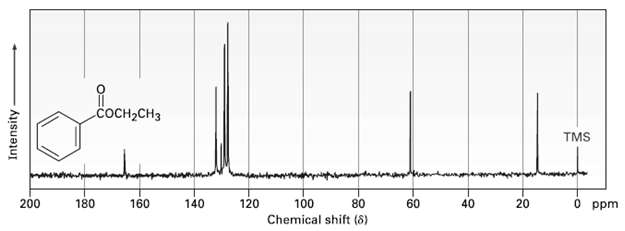
Transcribed Image Text:
соCH-CHз TMS 100 Chemical shift (8) 200 180 140 20 0 ppm 120 40 160 60 Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (18 reviews)
0 5 10 6 4 5 OO 2 ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
What changes would you expect in the 13C NMR spectrum of 1-bromopropane upon cooling the compound to very low temperature?
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
What is the difference between an optimistic approach and a pessimistic approach to decision making under assumed uncertainty?
-
American and European bricks-and-mortar retailing is increasingly becoming dominated by "hyper marts," enormous stores that sell groceries, household goods, hardware and other products under one...
-
For each of the following gas-phase reactions, write the rate expression in terms of the appearance of each product and disappearance of each reactant: (a) 2 H2O(g) -- 2 H2(g) + O2(g) (b) 2 SO2(g) +...
-
Define value realization and outline why realization is important for the entrepreneurial firm, the entrepreneur, and the community.
-
Mazurka Corporation has 2,000 shares of $100, 8 percent cumulative preferred stock outstanding and 40,000 shares of $1 par value common stock outstanding. In the companys first three years of...
-
Here are four major activities in drug law enforcement: Source Control - activities targeting cultivation, manufacturing and production of controlled dangerous substances in foreign countries....
-
Forty-five kilograms of a solution containing 30 wt% ethylene glycol in water is to be extracted with furfural. Using Figures 4.14a and 4.14c, calculate:(a) The minimum quantity of solvent.(b) The...
-
Long-range coupling between protons more than two carbon atoms apart is sometimes observed when ? bonds intervene. An example is found in 1-methoxy-1-buten-3-yne. Not only does the acetylenic proton,...
-
The 1H and 13C NMR spectra of compound A, C8H9Br are shown. Propose a structure for A, and assign peaks in the spectra to your structure. TMS O ppm 10 8. 6. Chemical shift (8) TMS 200 180 160 140 120...
-
Express each repeating decimal number as a quotient of two integers. If possible, reduce the quotient to lowest terms. 0.1
-
A zero-coupon bond with 8 years to maturity, trading for $941.73 and is expected to have it's annual YTM jump by 1% when the Federal Reserve announces its rate adjustments. What is the duration...
-
A bond with an annual coupon rate of 6.0% currently sells for $1,090. What is the bond's current yield?
-
What is the no-arbitrage price of a forward contract if the time to expiration is six months, the underlying asset is worth $250, the continuously compound annualized risk-free rate is 3%, and...
-
Green Life Corporation is trying to choose between two mutually exclusive projects. Project A has a 5-year expected life, an initial cost of $60,000 and a profitability index (PI) of 1.2. Project B...
-
Assume that capital markets are perfect. The price of a truck is $90,000, its residual value in four years will be $15,000, and there is no risk that the lessee will default on the lease. The...
-
If \(\vec{A}=A_{x} \hat{i}+A_{y} \hat{\jmath}\) and \(\vec{B}=B_{x} \hat{i}+B_{y} \hat{\jmath}\), show that \(\vec{A} \cdot \vec{B}=\) \(A_{x} B_{x}+A_{y} B_{y}\). You may wish to use the fact that...
-
Suppose that fraction used = / 1.0 + 0.1Mt. for some parameter 1. Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of ....
-
A KNO 3 solution containing 45 g of KNO 3 per 100.0 g of water is cooled from 40 C to 0 C. What happens during cooling?
-
Predict the products of the following reactions. (a) allyl bromide + cyclohexyl magnesium bromide (b) cyclopentadiene + anhydrous HCI (c) 2-methylpropene + NBS, light (d) Furan + trans 1,...
-
Show how the reaction of an allylic halide with a Grignard reagent might be used to synthesize the following hydrocarbons. (a) 5-methylhex-1-ene (b) 2,5,5-trimethylhept-2-ene (c)...
-
Draw the important resonance contributors for the following cations, anions, and radicals. (a) (b) (c) (d) (e) (f) (g) (h) CH CH 2 - CH2 OF OCH3
-
Stock #1 has a standard deviation of 55 percent. Stock #2 has a standard deviation of 75 percent. You invest 48 percent of your money in Stock #1 and the rest in Stock #2. If there were NO benefit to...
-
Eagle Industries' bonds have a 10-year maturity and a 8.00% coupon paid semiannually. They sell at their $1,000 par value, and are not callable. What s the effective annual rate (EFF %) for these...
-
Help Save & Ext The following transactions occur for the Panther Detective Agency during the month of July 1. Purchase a truck and sign a note payable, $13,900 2. Purchase office supplies for cash,...

Study smarter with the SolutionInn App


