Assign configuration to the following substance, and draw the structure of the product that would result on
Question:
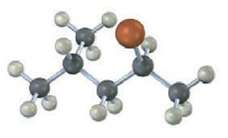
Assign configuration to the following substance, and draw the structure of the product that would result on nucleophilic substitution reaction with HS ? (reddish brown = Br):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
HS H CH3 B...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Draw the structure of the product from the following reaction (formed during a synthesis of one of the endiandric acids by K. C. Nicolaou): MeO2C osi(t-Bu)Phe toluene, 110C
-
Assign R or S configuration to the following molecule, write the product you would expect from SN2 reaction with NaCN, and assign R or S configuration to the product (yellow-green =Cl):
-
Consider the following set of data where an experiment was done to determine the molarity of a solute. What mass of sample is needed to reduce the %RSD to 0.10% if the sampling constant is 17.3 g?...
-
How could the construction company in the first example have met the legal requirements for overtime pay?
-
Some watch dials are coated with a phosphor, like ZnS, and a polymer in which some of the 1H atoms have been replaced by 1H atoms, tritium. The phosphor emits light when struck by the beta particle...
-
What sources does an auditor use to test for overstated accounts payable, and how are those sources used?
-
1. Imagine that an agency or business has asked for help in gathering data about the number of sports anglers who fish off the coast of Georgia. What advice would you give about sampling? What method...
-
In the case of the plucked string, use the formula for Fourier sine coefficients to show that Am 2h sin mp m p(x-p For what position of p are the second, fourth, ... harmonics missing? For what...
-
Tom Hruise was an entertainment executive who had a fatal accident on a film set. Tom's will directed his executor to distribute his cash and stock to his wife, Kaffie, the real estate to his church,...
-
What product would you expect to obtain from SN2 reaction of OH with (R)-2-bromo-butane? Show the stereo chemistry of both reactant and product.
-
What product would you expect from SN2 reaction of 1-bromobutane with each of the following? (a) NaI (b) KOH (c) H C C Li (d) NH3
-
Below are NMR spectra of several compounds. Identify whether these compounds are likely to contain ethyl, isopropyl, and/or tert-butyl groups: (a) (b) (c) (d) Proton NMR C3H12 Chemical Shift (ppm)...
-
The money manager of a R4 000 000 investment fund. The fund consists of four shares with the following investments and betas: Share Investment Beta Class A share R400 000 1.50 Class B share R600 000...
-
If a person at the beach is 6' tall and the wavelength is 8 feet, will this person be able to stand with his or her head above the water where the waves start to swell?? At what depth will the waves...
-
An issue of bonds with par of $1,000 matures in 8 years and pays 9% p.a. interest semi-annually. The market price of the bonds is $955 and your required rate of return is 8%. (a) Calculate the bonds...
-
When a horse pulls a 94 kg buggy down the road with a force of 27 N, the buggy hitch makes an angle of 35 degrees above the horizontal. Neglecting friction, what is the magnitude of the buggy's...
-
Trail Co. is expected to pay a cash dividend of $2.60 next year. This dividend is expected to grow at 5% to infinity. If you require a return of at least 9%, what is the most you should be willing to...
-
What are the rms and peak currents for a \(60 \mathrm{~W}\) bulb? (The rms voltage is the usual \(120 \mathrm{~V}\).) A. \(0.71 \mathrm{~A}, 0.71 \mathrm{~A}\) B. \(0.71 \mathrm{~A}, 0.50...
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
An aqueous solution is saturated in both potassium chlorate and carbon dioxide gas at room temperature. What happens when the solution is warmed to 85 C? a) Potassium chlorate precipitates out of...
-
Which of the following compounds are capable of being resolved into enantiomers? (a) N-ethyl-N-methylaniline (b) 2-methylpiperidine (c) 1-methylpiperidine (d) 1, 2, 2-trimethylaziridine (e) (f) (g)...
-
Complete the following proposed acid-base reactions, and predict whether the reactants or products are favored. (a) (b) (c) (d) + CH,COOH acetic acid pyndine +CH3COOH acetic acid pyrrole pyridinium...
-
Predict the products of the following reactions: (a) Excess NH3 + Ph - CH2CH2CH2Br (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q) (r) (I) NaN3 (2) LiAIH (3) H30 1-bromopentane CH3...
-
I am working in aMobile company that is seeking to introduce a novel mobile application that implements a monthly subscription fee for its users. After conducting a comprehensive analysis, the...
-
Tatiana has recently entered a profile into the Express Entry (EE) system as a candidate under the Canadian Experience Class (CEC). She has also met the requirements to qualify under the FSW. Tatiana...
-
Columbia Corp. is a young, rapidly growing company that manufactures computer accessories and specialized components for networked computer workstations. It has some unique products and a strong...

Study smarter with the SolutionInn App


